Spider Silk 5x Tougher Than Steel: Molecular Secret Cracked
Authors: Gregory P. Holland, Christian D. Lorenz, Hannah R. Johnson, Kevin Chalek

Spider silk’s specific strength exceeds that of steel, and its fracture toughness surpasses Kevlar by more than threefold (approximately 180 MJ/m³ vs. 50 MJ/m³). Yet, for a very long time, it remained entirely unclear how a spider manages to transform a liquid «soup» of proteins into one of the most robust threads on the planet in a fraction of a second. Now, researchers have finally unraveled this biochemical magic trick — and the discovery could forever change modern materials manufacturing.
Historical Context
Spider silk is biodegradable, lighter than nylon, and stronger than steel per unit weight. Engineers want it for medical implants, body armor, suspension-bridge cables — basically anything that needs to be tough and light at once.
However, all attempts at the mass production of artificial spider silk have met with failure. Farming spiders, akin to silkworms, is impossible because spiders are highly territorial and prone to cannibalism. While scientists learned to synthesize the core proteins (spidroins) in the lab using genetically modified bacteria, pulling a thread from them resulted in a brittle and weak material. The problem did not lie in the composition of the synthesized «syrup, ” but in the mechanism of its crystallization. Scientists lacked an understanding of what precisely triggers the structural solidification at the molecular level.
In 2018, Gregory Holland’s laboratory at San Diego State University (SDSU) first explained how spiders safely store silk proteins in their glands without premature aggregation. But the critical question remained: what exactly triggers the transition from a liquid state to a solid fiber?
The Crucial Discovery
An international team led by Professor Gregory P. Holland (San Diego State University) and Professor Christian D. Lorenz (King’s College London) has finally identified the «missing link.» It turns out that the «magic button» activating solidification is a common inorganic ion — phosphate.
The results were published on December 22, 2025, in Proceedings of the National Academy of Sciences (PNAS, Volume 122, Issue 52, DOI: 10.1073/pnas.2523198122). The research was funded by the U.S. Air Force Office of Scientific Research (AFOSR), reflecting the strategic interest in materials of the future.
Cation-π interactions A unique type of non-covalent but exceptionally strong chemical bond. The positively charged guanidinium group of arginine is attracted to the electron-rich benzene ring of tyrosine, binding the molecules together like a heavy-duty microscopic Velcro fastener.
Researchers found that the addition of phosphate displaces water molecules surrounding the silk proteins, critically reducing their solubility. Phosphate acts as a trigger, forcing the amino acids arginine and tyrosine to seek each other out and bond via cation–π interactions.

How It Works
To achieve their breakthrough, the team employed a combination of cutting-edge methods: AlphaFold3 protein structure prediction, molecular dynamics simulations, and NMR spectroscopy (Nuclear Magnetic Resonance).
During the early stages of silk formation, as the solution moves through the spider’s gland duct, the concentration of phosphate increases. Phosphate ions displace water molecules, removing the «hydration barrier» between proteins. The proteins form droplets, undergoing phase separation — they separate from the water much like drops of oil. At this precise moment, arginine and tyrosine «snap» together.
Subsequently, when the spider mechanically draws out the thread, these micro-droplets (now locked together by chemical bonds) organize into an elongated, dense β-sheet structure. Crucially, arginine becomes partially incorporated into the crystalline regions (β-sheets), while tyrosine frequently forms β-turns at the boundaries between ordered and amorphous regions. This arrangement creates a nanostructural composite that gives the fiber both stiffness and elasticity simultaneously.
Why This Changes Everything
Knowing how to activate this process using a simple phosphate buffer paves a direct path to the scalable, industrial synthesis of spider silk. It is now possible to create liquid spidroins in vats and then, by controllably altering the chemical environment (adding phosphate), produce fiber through artificial spinning methods.
Ultra-lightweight aerospace skins, eco-friendly plastic alternatives, highly biocompatible surgical sutures — all of these move from «interesting lab demo» to «plausible product.» The synthetic spider silk market is already valued at $12.4 billion and is projected to reach $20 billion by 2035.
Surprisingly, the biochemistry of this process shares parallels with neurodegenerative diseases. The formation of amyloid plaques in Alzheimer’s disease occurs through remarkably similar protein phase separation — the same cation-π interactions have been found in human neurotransmitter receptors. But while spiders have evolutionarily refined their control over this process, in the brain it goes haywire. Studying spiders may provide crucial clues for understanding and treating brain diseases.
A Critical Look
The paper has been peer-reviewed and published in PNAS (December 2025).
What makes this study particularly convincing is the methodological rigor behind a deceptively simple answer. The identification of phosphate as the molecular trigger isn’t a speculation — it’s a conclusion reached independently through AlphaFold3 structural prediction, molecular dynamics simulations, and NMR spectroscopy. Three distinct methods pointing at the same mechanism is about as close to scientific certainty as you can get at this scale. The work also doesn’t appear from nowhere: it builds directly on Holland’s 2018 findings about how spiders prevent premature aggregation in their glands, finally closing the loop on a question that has frustrated materials scientists for decades. And the reach extends well beyond textiles — the same cation-π interactions implicated here appear in the aggregation pathways of amyloid proteins, quietly suggesting that spider biology may inform how we understand neurodegenerative disease.
That said, knowing the trigger and controlling it industrially are very different problems. The spider’s spinning duct is a remarkably precise microfluidic device that maintains finely tuned gradients of pressure, pH, and ion concentration along its length — conditions that no artificial spinneret has yet convincingly replicated. Understanding that phosphate initiates phase separation is a necessary precondition for synthetic silk production, but it is not a sufficient one. The economic viability of scaling phosphate-induced synthesis from a laboratory vat to a commercially competitive process remains entirely undemonstrated.
The most pressing open question is therefore one of engineering rather than biochemistry: how precisely must the rate and spatial gradient of phosphate concentration be controlled within an artificial spinneret to produce threads with consistent mechanical properties at commercial scale? The molecular instructions are now decoded. Building the machine that follows them is the next chapter.
What’s Next
The next frontier is the creation of commercially viable artificial spinning rigs that utilize the phosphate trigger. According to Gregory Holland, previous attempts at silk synthesis failed precisely because researchers replicated the «ingredients» (proteins) but not the «instructions» (the molecular assembly mechanism). Now, those instructions have finally been decoded.
References
Related Articles
Muscle Knots: 2024 Trigger Point Breakthrough
Why muscle knots form, how they harm your health, and how to treat them — featuring the molecular pathway discovered in 2024.
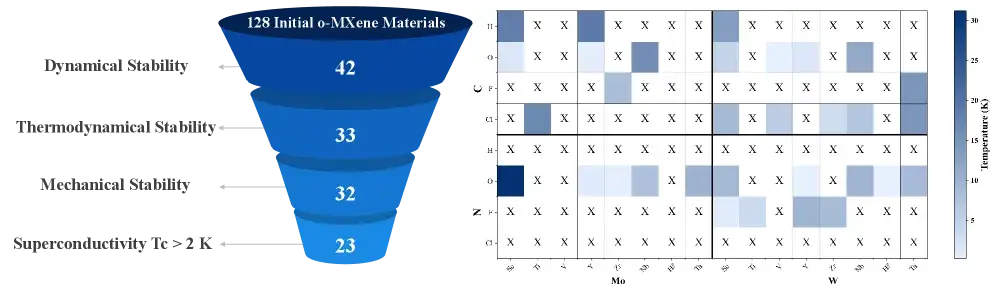
52 K Superconductor: 2D MXenes Breakthrough
Scientists predicted superconductivity at 52 K in functionalized MXenes — the highest predicted temperature for any 2D material.
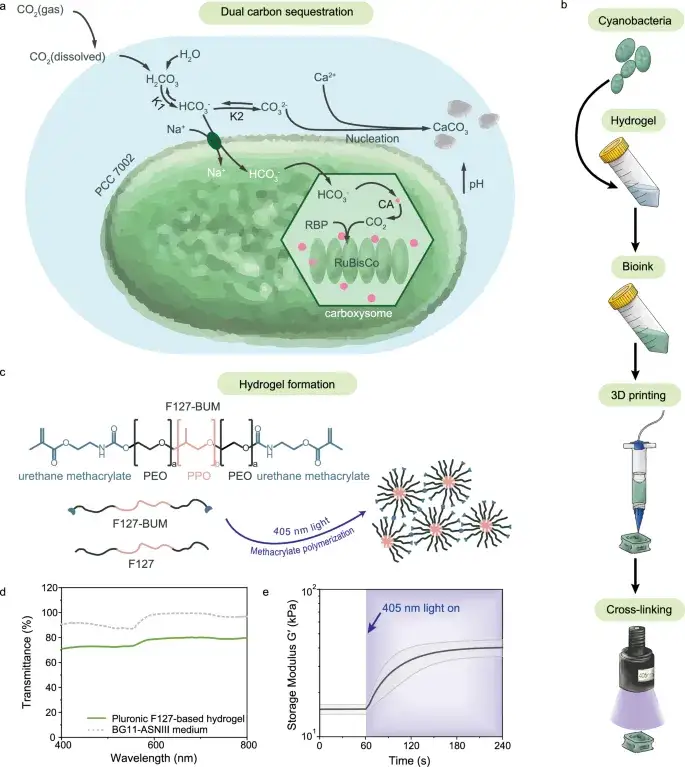
Bacteria Gel Captures CO₂ While Growing Stronger (2025)
ETH Zurich engineered a 3D-printable hydrogel packed with cyanobacteria. It absorbs CO₂ via photosynthesis and grows harder over time.