How Sleep Loss Turns Your Gut Against Your Brain
Authors: Na Zhao, Jingqi Sun, Ronghua Xu, Yafei Wang, Hao Yu, Ke An, Wenjie Zhang, Bo Jiang, Fang Xie
Three sleepless nights. Your head throbs, coffee stopped working hours ago, and the world feels slightly hostile. But the real damage is not happening behind your eyes. It is unfolding several feet lower — in your gut. Your intestinal bacteria have already shifted. A microbial balance maintained for years collapses in days. And the broken gut starts a slow, methodical assault on your brain.
A team of Chinese neuroscientists led by Fang Xie at the Third Military Medical University in Chongqing has mapped this assault in granular detail. They traced a nine-step molecular chain that begins with disrupted sleep and ends with tau tangles — the hallmark lesion of Alzheimer’s disease.
From Colon to Cortex: The Nine-Link Chain
The most striking moment of this study is not the chain itself. It is a single transplant experiment.
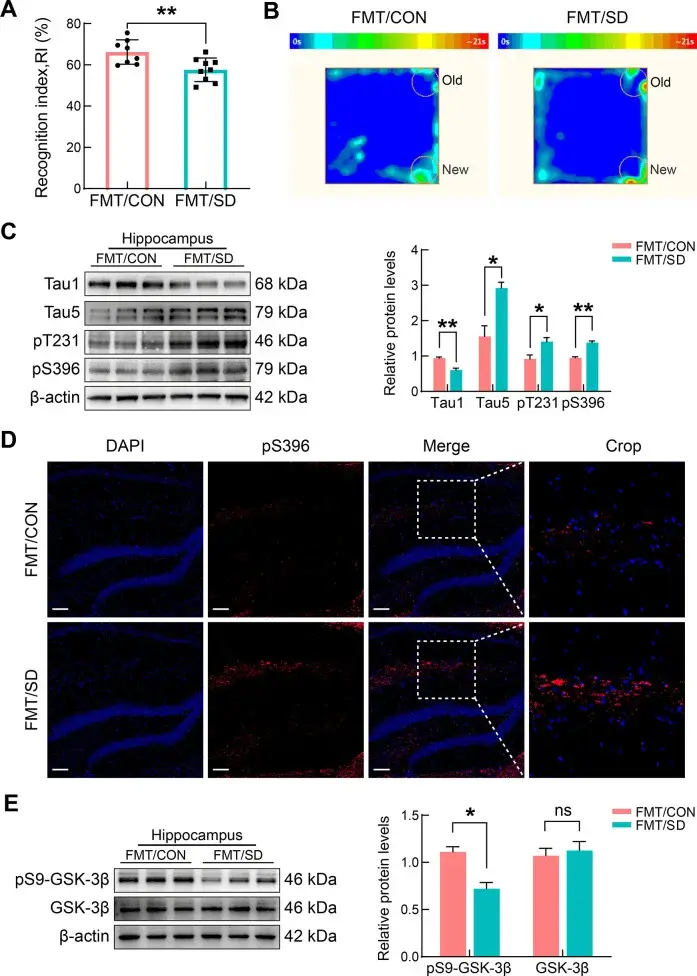
The researchers collected gut bacteria from chronically sleep-deprived mice and transferred them into healthy, well-rested animals. No stress. No sleep restriction. Just foreign microbes. Within weeks, the recipients developed the same brain damage: hyperphosphorylated tau protein, neuroinflammation, crippled autophagy.
Autophagy — literally «self-eating.» A cellular housekeeping process where the cell wraps damaged proteins and organelles into membrane vesicles and digests them. When autophagy fails, molecular garbage piles up — and neurons begin to die.
That transplant is the linchpin. It shows that sleep deprivation does not attack the brain directly. It attacks through the gut. The intestine is not a bystander. It is the relay station.
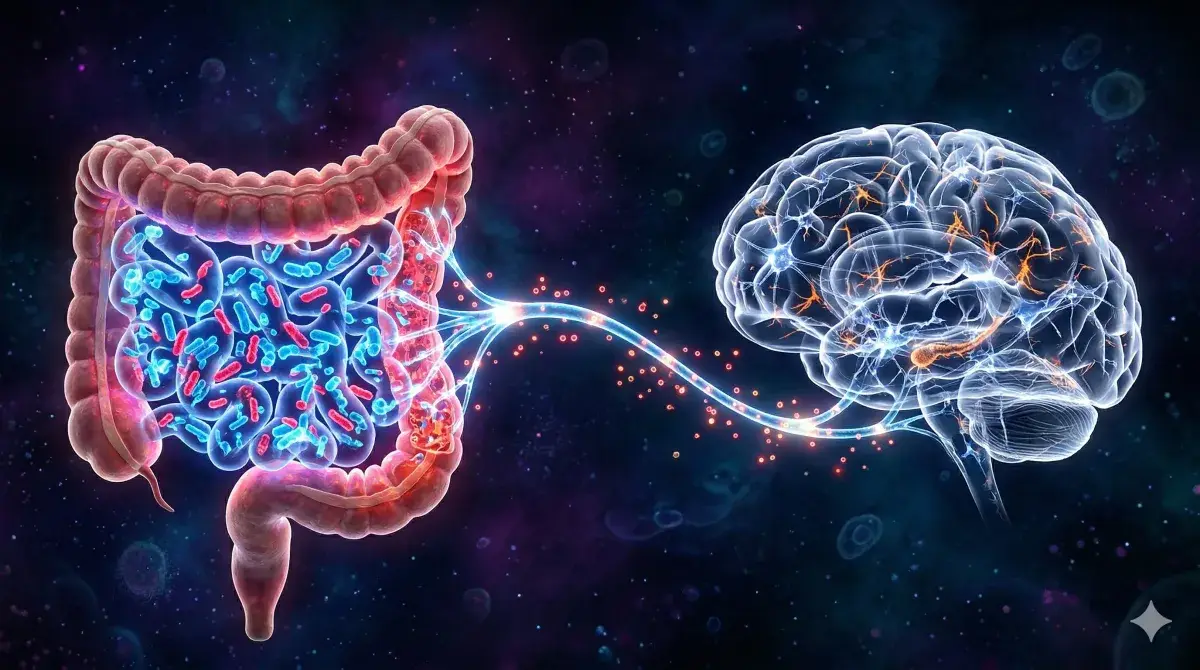
Here is how the full chain unfolds. Chronic sleep loss reshapes the gut microbiota: protective species like Lactobacillus retreat while pro-inflammatory taxa expand. The damaged microbiota weakens the intestinal barrier — the single-cell wall that normally blocks bacteria from reaching the bloodstream. Bacterial molecules leak through.
Intestinal barrier — a one-cell-thick lining inside the gut that selectively absorbs nutrients while blocking bacteria. When breached, bacterial components enter the blood and trigger systemic inflammation.
Once bacterial signals reach the brain, they activate the NLRP3 inflammasome in microglia — the brain’s resident immune cells.
When Your Immune System Turns on Your Brain
NLRP3 inflammasome — a multi-protein complex inside immune cells that functions as a danger detector. When it senses threats (infection, tissue damage, toxic protein aggregates), it assembles and triggers the release of inflammatory cytokines IL-1β and IL-18. The problem: chronic activation means the fire brigade itself starts burning the building down.
The activated inflammasome does more than trigger inflammation. It suppresses autophagy by phosphorylating Beclin-1, a master regulator of the autophagic process. With Beclin-1 locked, the cellular cleanup system grinds to a halt.
At the same time, the enzyme GSK-3β enters the picture. This protein plays a remarkable dual role: it simultaneously amplifies NLRP3 inflammasome activity (boosting inflammation) and phosphorylates tau protein (seeding the tangles that define Alzheimer’s pathology). One enzyme, two strikes.
With autophagy offline and GSK-3β running unchecked, hyperphosphorylated tau accumulates in neurons. Nobody clears it — the cleanup crew is disabled. Tangles build. Neurons start dying.
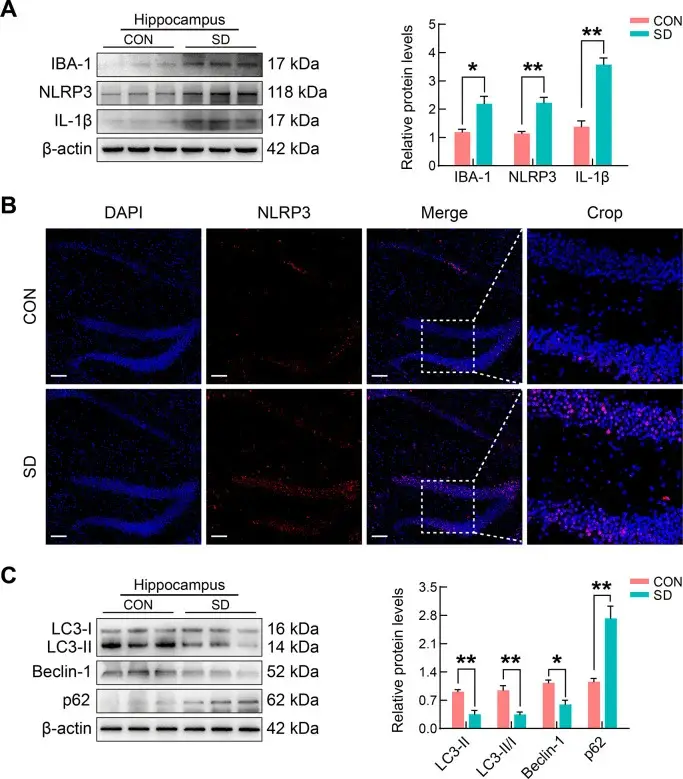
The Recycling System That Shut Down
In a healthy brain, autophagy is a powerful defense. It continuously breaks down defective proteins — including tau aggregates and amyloid-beta. Think of it as a garbage truck that runs every street, every day.
Now imagine the truck stops. Trash bags pile up on Monday. By Friday the sidewalk is impassable. By the end of the month, the neighborhood is uninhabitable. That is what happens to neurons when Beclin-1 is blocked: tau is no longer recycled, so it aggregates, clumps, and kills the cell.
Xie’s team confirmed this experimentally. When mice with disrupted microbiota received rapamycin — a drug that stimulates autophagy — tau pathology was significantly reduced. The garbage truck was back on its route.
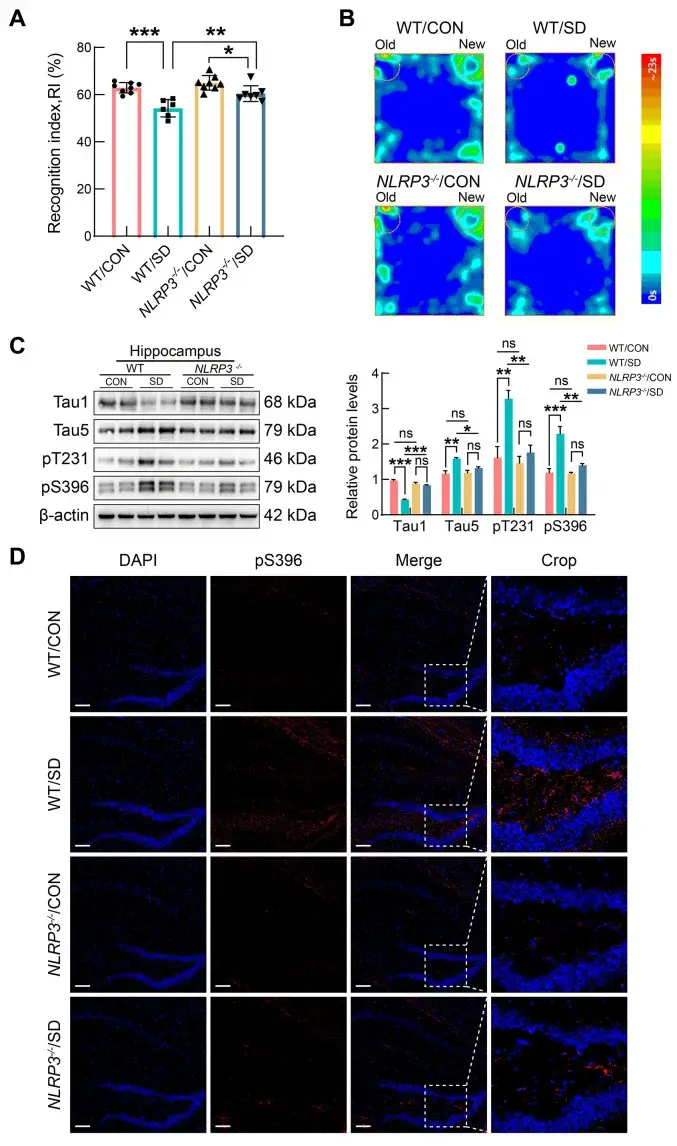
Two Sleepless Nights Were Enough
The mouse model used 20 hours of wakefulness per day over several weeks. That is extreme. But what about humans?
Human data remain fragmentary, yet concerning. Christian Benedict at Uppsala University showed in 2016 that two sleepless nights sufficed to alter gut microbiota composition and epigenetic marks in adipose tissue. A 2017 study by Zhang and colleagues documented microbial shifts after five nights restricted to four hours of sleep.
No one has yet traced the full nine-link chain in humans — from microbiota through NLRP3 to tau. But every individual link has been confirmed in human studies: sleep loss alters the microbiome; increased gut permeability correlates with neuroinflammation; NLRP3 is activated in Alzheimer’s brains; impaired autophagy appears in postmortem brain samples from patients with the disease.
What This Study Cannot Tell Us
The study was published in Zoological Research (July 2024) and has undergone peer review.
The fecal microbiota transplant experiment is the strongest argument for causation. It elevates this work above the typical correlational studies that flood gut-brain literature. Instead of «A is associated with B, ” we get „transferring A causes B.“
Yet a mouse brain is not a human brain. Mice do not develop classical Alzheimer’s disease: they lack the natural accumulation of amyloid plaques and neurofibrillary tangles that define the condition. The tau pathology observed here is a model, not a diagnosis. Direct clinical translation requires prospective cohort studies tracking sleep, microbiome, and cognitive status in parallel over years.
The sleep deprivation protocol itself is another caveat. Twenty hours of daily wakefulness is an extreme regimen — rare even among the most relentless workaholics. How the chain behaves under moderate chronic deficit — say, six hours of sleep instead of eight over several years — remains an open question.
There is also a methodological nuance: the transplant experiment does not rule out the possibility that metabolites, phages, or other fecal components — not bacteria alone — drive the cascade. Identifying the precise trigger within the transplanted material is a task for future work.
The Road Ahead
If the gut is the relay between sleep and the brain, it can theoretically be targeted from two directions. From the sleep side — obviously: sleep more and sleep better. From the gut side — through probiotics, diet, and physical activity.
A 2025 randomized placebo-controlled trial tested a multi-strain probiotic supplement in healthy older adults over 12 weeks. The results showed improved episodic memory scores and reduced systemic inflammation markers. Modest, but statistically significant. Not a cure for Alzheimer’s — but perhaps one brick in the wall of prevention.
Exercise attacks the same axis from a different angle. A 2023 meta-analysis found that regular aerobic activity increases gut microbial diversity and reduces neuroinflammation markers. The mechanism likely overlaps with what Xie’s team described: a healthy microbiome keeps NLRP3 quiet, autophagy running, and tau aggregates in check.
The central question ahead: can we catch the chain early and break it while damage is still reversible? The answer will determine whether the sleep-gut-brain axis becomes a real therapeutic target or remains an elegant diagram in neuroscience textbooks.
Frequently Asked Questions
Can poor sleep really cause Alzheimer’s disease?
A direct causal link in humans has not been proven. However, epidemiological data consistently show that chronic sleep deficiency is associated with increased dementia risk. The Xie study is the first to describe a complete molecular chain — from sleep loss through the gut to tau pathology — but so far only in mice.
Could probiotics protect the brain from sleep deprivation damage?
Early clinical evidence is encouraging: a 12-week probiotic course improved memory in older adults in a 2025 randomized trial. But probiotics have not been specifically tested for protecting the brain against sleep loss consequences. It is a promising direction, though recommending specific strains would be premature.
How quickly does sleep loss affect the gut?
In humans, changes in gut microbiota composition have been detected after just two sleepless nights. Five nights restricted to four hours of sleep produce even more pronounced shifts. How quickly these changes become clinically significant remains unknown.
What is the gut-brain axis and why does it matter?
It is a bidirectional communication channel between the intestinal microbiota and the central nervous system. It operates through the vagus nerve, immune signaling molecules, and bacterial metabolites. Disrupted microbial balance can influence brain inflammation, mood, and cognitive function. The Xie study showed this channel can also transmit neurodegenerative signals.
Can brain damage be reversed by getting more sleep?
In the mouse experiment, stimulating autophagy with rapamycin reduced tau pathology, suggesting some reversibility. But human data on whether damage reverses after sleep normalization do not exist. Early-stage disruption is likely recoverable, but prolonged chronic sleep deprivation may cause permanent harm.
References
Related
- Acute sleep loss results in tissue-specific alterations in genome-wide DNA methylation state and metabolic fuel utilization in man (Science Advances)
- Sleep Deprivation and Gut Microbiota Dysbiosis: Current Understandings and Implications (Int. J. Mol. Sci.)
- Effects of a probiotic supplement on cognitive function in healthy older adults: a 12-week RCT (Nutrients, 2025)
Related Articles
Coffee vs. Dementia: Harvard Tracked 132,000 People for 40 Years
The largest observational study of its kind in JAMA: 2–3 cups of coffee or 1–2 cups of tea per day linked to 18% lower dementia risk. Decaf showed no benefit — and that tells us something important.
Bacteria That Eat Tumors: Cancer Therapy Breakthrough
Engineers gave Clostridium sporogenes quorum sensing — bacteria find tumors, wait for backup, then destroy cancer from within.
A Bacterium Frozen for 5,000 Years Is Resistant to 10 Antibiotics — and May Help Discover New Ones
A 5,000-year-old microbe from a Romanian ice cave resists 10 modern antibiotics. Its genes may hold keys to discovering new drugs.