Muscle Knots: 2024 Trigger Point Breakthrough
Authors: Yu Liu, Feng Qi
Why It Matters
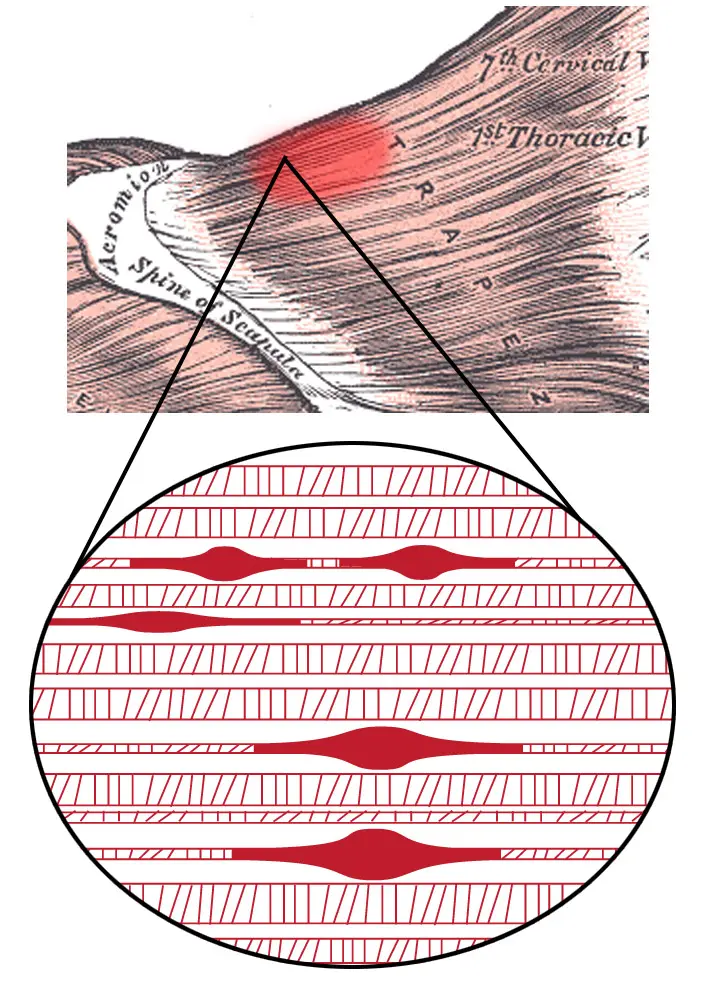
Anyone who has ever felt their neck after a long day at the desk knows the sensation: a hard, painful lump somewhere between the shoulder blade and the spine. Press on it, and the pain radiates to your temple, arm, or down your back. This is what people call a «muscle knot» — and what medicine calls a myofascial trigger point.
By some estimates, myofascial pain syndrome affects up to 30% of the world’s population. It ranks among the most common causes of chronic neck, back, and shoulder pain. Yet scientists still debate what these «knots» actually are and why they form.
In 2024, a team from Shandong University published a paper in Anesthesiology that described, for the first time, a specific molecular mechanism behind muscle knot formation. The discovery of the COL1A1/PDGFR-α/JAK2/STAT3 axis is not just another hypothesis — it is an experimentally validated pathway from molecule to pain. And potentially, a key to fundamentally new treatments.
The Classic Theory: Energy Crisis
To appreciate the 2024 breakthrough, we first need to understand what we «knew» before.
Myofascial trigger point (MTrP) — a hyperirritable nodule within a taut band of skeletal muscle. When pressed, it produces local pain, referred pain in distant areas, and sometimes an involuntary twitch of the muscle bundle.
The classic «integrated hypothesis» by David Simons, formulated in the 1990s, describes a muscle knot as a vicious cycle:
- Initiating event — microtrauma, overload, prolonged static tension
- Acetylcholine excess — the neuromuscular junction releases too much neurotransmitter
- Stuck contraction — several sarcomeres lock into a shortened state
- Ischemia — the spasming area compresses capillaries, reducing blood flow
- Energy crisis — without oxygen, ATP synthesis drops; without ATP, the calcium pump cannot relax the muscle fiber
- Pain — hypoxia and tissue damage release a «cocktail» of pain mediators: substance P, bradykinin, prostaglandins, cytokines
Sarcomere — the smallest contractile unit of a muscle. Imagine two sets of combs interlocked with each other. When the muscle contracts, these «combs» — the proteins actin and myosin — slide past one another, shortening the sarcomere. To slide apart again, they need energy in the form of ATP.
The model is elegant and logical. But here is the problem: it describes a closed loop without explaining what exactly, at the molecular level, initiates and sustains this state. Electrophysiologists have recorded spontaneous electrical activity in trigger point zones for decades — but what drives it?
The Breakthrough: COL1A1 → PDGFR-α → JAK2/STAT3
In July 2024, Yu Liu and colleagues from Shandong University asked a simple question that, surprisingly, had not been investigated before: if a trigger point is a pathological tissue process, which receptor tyrosine kinases are activated in it?
Receptor tyrosine kinases (RTKs) — protein receptors on cell surfaces that, when activated, trigger cascades of intracellular signals. Think of them as «antennas» receiving molecular messages. PDGFR-α is one such receptor, typically associated with cell growth and wound healing.
The researchers biopsied the upper trapezius muscle from 11 patients with chronic myofascial pain and 7 healthy volunteers. Using an RTK phosphorylation microarray, they screened dozens of receptors simultaneously — and found that one stood out dramatically from the rest.
PDGFR-α — platelet-derived growth factor receptor alpha. Its activation in trigger point tissues was significantly elevated (p < 0.001). Moreover, the level of activated PDGFR-α correlated with pain intensity on the visual analog scale (r = 0.711). The worse the pain, the more phosphorylated receptor in the tissue.
But what activates this receptor? The classic PDGFR-α ligand is PDGF-AA. Its blood levels in patients were also elevated (5.97 ng/ml vs. 3.74 in controls). However, the real surprise came next.
Collagen as a Signaling Molecule
Using mass spectrometry and co-immunoprecipitation, the authors discovered that PDGFR-α physically binds to COL1A1 — collagen type I, the primary structural protein of connective tissue.
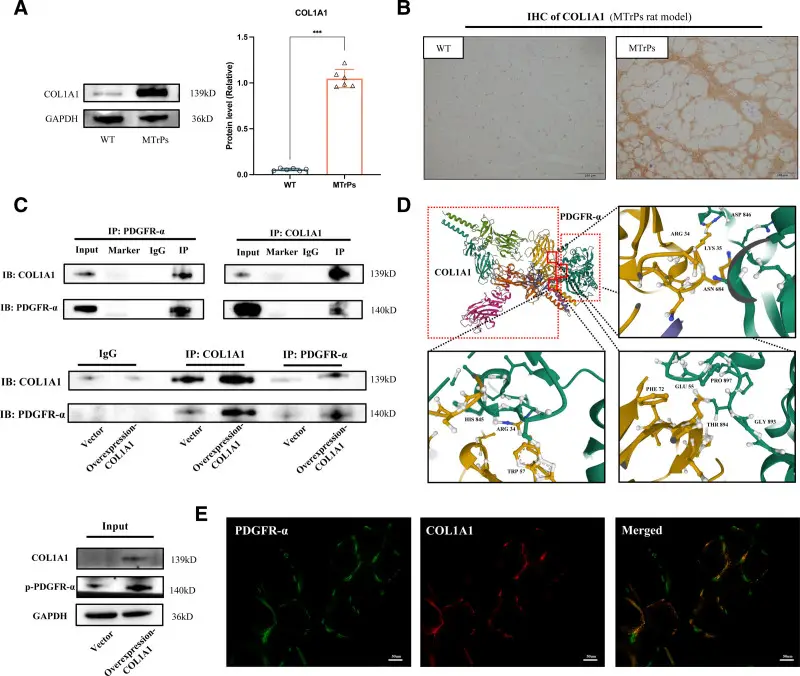
This was unexpected. Collagen I is a «scaffolding» protein that everyone considered a passive structure — essentially, the rebar of tissues. But it turns out COL1A1 also functions as a signaling molecule: it binds PDGFR-α and activates it directly, without the classical PDGF ligand.
Molecular docking revealed specific binding sites: amino acids LYS35–ASP846, GLU55–THR894, HIS845–ARG34.
From Molecule to Pain
The authors then validated the entire chain in a rat model. The cascade begins with COL1A1 accumulating in damaged muscle areas — from microtrauma, overload, or chronic tension. This excess collagen binds directly to PDGFR-α, triggering its phosphorylation, which in turn engages the JAK2/STAT3 signaling pathway — a classic inflammatory cascade. Two things happen simultaneously downstream: pro-inflammatory cytokines (IL-1β, IL-6, TNF-α) flood the local tissue, and myosin light chain kinase (MLCK) phosphorylates myosin, locking sarcomeres into sustained contraction. The fibers shorten from their normal 2.13 μm to just 1.63 μm. The end product is a contraction knot — a self-reinforcing loop of spasm, inflammation, and pain.
JAK2/STAT3 — one of the key intracellular signaling pathways. JAK2 is a kinase enzyme that, when activated, phosphorylates the transcription factor STAT3. Activated STAT3 enters the cell nucleus and switches on genes for inflammation and contraction. This pathway is well known in oncology — and now it turns out to be involved in muscle pain.
The critical finding: when the researchers blocked this pathway — either by knocking down PDGFR-α or by using the JAK2/STAT3 inhibitor AZD1480 — pain behavior in rats decreased, sarcomeres returned to normal length, and inflammatory cytokine levels dropped.
Why Muscle Knots Are Harmful
«Just some neck tension, ” someone might say. But myofascial trigger points are more than mere discomfort — they set off a chain of consequences that extends far beyond the original sore spot.
The most insidious is pain chronification. A constant barrage of signals from trigger points rewires the spinal cord through a process called central sensitization: the nervous system gradually turns up its sensitivity until stimuli that used to go unnoticed begin to hurt. Clinicians call this allodynia, and it means the threshold for pain keeps dropping long after the initial injury has healed.
Worse, a single trigger point rarely stays alone. It tends to activate satellite points in neighboring muscles, antagonist muscles, and sometimes areas far from the original site. A patient who walks in with neck pain may discover painful lumps in their lower back and thighs six months later — not a new injury, but the original problem spreading through muscular chains.
Then come the secondary effects: chronic pain wrecks sleep architecture, poor sleep amplifies pain sensitivity, and both raise the risk of anxiety and depression — which in turn increase muscle tension, creating three interlocking vicious cycles. Meanwhile, the affected muscles lose range of motion, force the body into guarding postures, and grow weaker. Studies on the trapezius show that trigger points there significantly restrict neck mobility and increase the incidence of chronic tension-type headaches.
The Scientific Debate: Do Trigger Points Even Exist?
Trigger points remain one of the most contested topics in medicine — and the debate is fiercer than you might expect.
Skeptics, led by John Quintner from the IASP (International Association for the Study of Pain), point to serious issues:
- Different clinicians palpating the same patient often locate trigger points in different places
- Treatment effects are difficult to distinguish from placebo
- Diagnosis relies on circular logic: «it hurts → found a tender spot → therefore the spot causes pain»
Proponents counter that:
- Biochemical changes in trigger point zones are reproducible — substance P, cytokines, local acidosis
- 2024 ultrasound elastography showed trigger points are objectively stiffer than surrounding tissue, with measurable parameters
- The Liu et al. study provides the first concrete molecular mechanism
The modern compromise (as of 2024–2025): painful muscle lumps are a real clinical phenomenon. But they are probably not a separate «disease» — rather, a combination of local muscular changes and overall nervous system sensitivity. The PDGFR-α work bolsters the «peripheral» component, but the role of central sensitization in chronification remains crucial.
How to Get Rid of Them: What the Evidence Says
Systematic reviews from 2024–2025 allow us to rank methods by strength of evidence:
Dry needling — the most studied method. Meta-analyses show moderate-to-strong short-term pain reduction and increased pain thresholds. Effects last up to 6 weeks. Requires a trained practitioner.
Dry needling — insertion of a thin needle directly into the trigger point. Unlike acupuncture, there are no «meridians» — the needle targets the spasm zone specifically, provoking a local twitch response followed by muscle relaxation.
Massage and manual therapy — moderate evidence. Friction massage of trigger points raises pain thresholds and increases range of motion. Multiple sessions work better than one. Safe, accessible, pairs well with exercise.
Extracorporeal shockwave therapy (ESWT) — emerging evidence for trigger points in the lower back. Evidence quality remains low to moderate.
Foam rolling and self-massage — reduces soreness and improves mobility in the short term. But rigorous data specifically on clinical trigger points are sparse. Cheap, safe, and convenient for daily use.
Exercise — the cornerstone of any protocol. Stretching + strengthening target muscles + ergonomic correction. Without this, all other methods deliver only temporary relief.
Trigger point injections — a 2023 meta-analysis showed moderate advantage over medication for acute myofascial pain. Practitioners use lidocaine, botulinum toxin, or even just saline — interestingly, the «dry needle» effect is often comparable to injection.
Critical Assessment
The study’s greatest methodological strength is that it did not start from a pre-chosen hypothesis. Instead of testing whether, say, substance P or bradykinin was elevated, the team ran an unbiased RTK phosphorylation microarray across dozens of receptors simultaneously — and let the data point to PDGFR-α. That agnostic design makes the finding considerably harder to dismiss as confirmation bias. Equally important is the causal architecture of the evidence: the authors did not stop at correlation. They knocked down PDGFR-α genetically and blocked JAK2/STAT3 pharmacologically with AZD1480, and in both cases pain behavior decreased, sarcomere length normalized, and inflammatory cytokine levels dropped. The biopsy-based approach — comparing active trigger point tissue with healthy muscle from the same anatomical region — also controls for many between-subject confounds that plague pain research.
That said, the human data rests on a fragile foundation: 11 patients and 7 controls recruited at a single university hospital. A sample that size can establish a signal but cannot determine whether the COL1A1/PDGFR-α axis is a universal mechanism for all myofascial trigger points or merely one pathway among several, relevant only to a specific subset of patients or injury types. The animal model carries its own limitations — blunt trauma to the rat gastrocnemius is a practical way to induce contraction knots, but it only approximates the chronic, low-load overuse patterns that produce most human trigger points. Translating rodent pain studies to human chronic pain has a decidedly mixed track record, and the pathway’s behavioral relevance in humans remains to be established in independent cohorts.
The most pressing open question is mechanistic rather than clinical: how does this newly described molecular cascade interact with the treatments that already work? Dry needling, massage, and exercise all demonstrably reduce trigger point pain, but none of them were designed with PDGFR-α in mind. Understanding whether these interventions downregulate COL1A1 accumulation, interrupt JAK2/STAT3 signaling, or act through entirely separate routes would significantly sharpen both the science and the therapy. As for PDGFR-α or JAK2/STAT3 inhibitors as a direct treatment — existing JAK inhibitors like ruxolitinib are already approved in rheumatology and oncology, which shortens the preclinical distance, but local administration for muscle pain would still require years of safety and dosing trials before reaching patients.
The study was published in peer-reviewed Anesthesiology (IF ~9), though independent replication of these findings is still needed. This article is a popular science overview, not medical advice.
What Comes Next
The discovery of the COL1A1/PDGFR-α/JAK2/STAT3 axis opens several avenues:
New therapeutic targets. JAK2 inhibitors already exist — ruxolitinib, baricitinib, tofacitinib. They are used in rheumatology and oncology. Theoretically, local injection of such drugs into the trigger point zone could be more effective than existing injections. But this remains pure hypothesis for now.
Objective diagnostics. If phosphorylated PDGFR-α levels correlate with pain intensity, this could become the first objective biomarker of myofascial pain — replacing subjective palpation.
Bridging the camps. The Liu et al. data could reconcile trigger point skeptics and proponents: there is an objective molecular substrate, but it is tightly linked to neuroinflammation and central sensitization. Both sides of the debate are partially right.
In the meantime, if your neck hurts right now — stand up, stretch, move your shoulders. Exercise is the only method with zero cost and zero side effects that works whether you believe in «mysterious trigger points» or «just tense muscles.» Whichever camp turns out to be right.
Frequently Asked Questions
Are muscle knots the same as muscle spasms?
No. A muscle spasm is an involuntary contraction of an entire muscle or a large portion of it, usually brief. A muscle knot is a sustained contraction of just a few sarcomeres within a single muscle bundle. It can persist for weeks or months, unlike a spasm that typically resolves within minutes.
Can I identify a trigger point on my own?
Typical signs during self-palpation: a firm nodule in the muscle, sharp tenderness when pressed, and pain that radiates to another area — for instance, from the neck to the temple. However, research shows that even professionals do not always agree when palpating the same patient. If the pain is chronic or significantly affects your quality of life, consult a specialist rather than relying on self-diagnosis.
Does stress really cause muscle knots?
Yes, the link is supported by research. Psychological stress raises muscle tone via sympathetic nervous system activation, impairs microcirculation, and lowers pain thresholds. During exam periods, for example, students show significantly more active trigger points in the trapezius muscle. But stress is more of a perpetuating factor than the sole cause.
How effective is foam rolling against trigger points?
Foam rolling moderately reduces muscle soreness and improves mobility — systematic reviews confirm this. However, rigorous data specifically on clinical trigger points remain limited. A foam roller is a solid daily prevention tool, but for persistent chronic pain, it is unlikely to replace targeted massage, dry needling, or an exercise program.
When can we expect new drugs based on the PDGFR-α discovery?
Not anytime soon. The Liu et al. findings are basic science: they demonstrate a molecular mechanism in rats and a small group of patients. Years of additional research are needed before clinical trials of targeted therapy — validation in larger cohorts, safety assessment of local JAK2 inhibitor administration, and dosing protocol development. An optimistic estimate is 5–10 years before the first clinical protocols emerge.
References
Related
- Advancing musculoskeletal diagnosis and therapy: a comprehensive review of trigger point theory and muscle pain patterns
- Myofascial Pain Syndrome: An Update on Clinical Characteristics, Etiopathogenesis, Diagnosis, and Treatment
- Myofascial trigger points: spontaneous electrical activity and its consequences for pain induction and propagation
- Mechanisms of Myofascial Pain
- Myofascial trigger point (MTrP) size and elasticity properties can be used to differentiate characteristics of MTrPs in lower back skeletal muscle
- Treating myofascial pain with dry needling: a systematic review
- The effect of friction massage on pain intensity, PPT, and ROM in individuals with myofascial trigger points
Related Articles
Bacteria That Eat Tumors: Cancer Therapy
Engineers gave Clostridium sporogenes quorum sensing — bacteria find tumors, wait for backup, then destroy cancer from within.
Big Five Personality: 254 Genes Found, Sixth Factor (2025)
Genome study of 600K people found 254 genes shaping personality. A 6th trait beyond Big Five predicts mortality. Seven 2024-2025 studies reviewed.
Spider Silk 5x Tougher Than Steel: Molecular Secret Cracked
Scientists decoded the exact molecular switch — arginine-tyrosine bonds — that turns liquid protein into spider silk, a thread 5x tougher than steel.