Bacteria Gel Captures CO₂ While Growing Stronger (2025)
Authors: D. Dranseike, Y. Cui, A. S. Ling, F. Donat, S. Bernhard, M. Bernero, A. Areeckal, M. Lazic, X.-H. Qin, J. S. Oakey, B. Dillenburger, A. R. Studart, M. W. Tibbitt
Why It Matters
Concrete is the second most consumed material on Earth after water. And one of the largest sources of CO₂ emissions: the cement industry accounts for roughly 8% of global carbon dioxide emissions. Billions of tons of CO₂ are released into the atmosphere every year so we can build homes, bridges, and roads.
What if a building material not only didn’t emit carbon dioxide, but actively absorbed it? Continuously — for as long as the building stands?
A team of 13 researchers led by Mark Tibbitt at ETH Zurich has created exactly that. It’s a transparent 3D-printable hydrogel harboring living cyanobacteria — among the oldest photosynthetic organisms on Earth. The material captures CO₂ through two simultaneous pathways: photosynthesis (like plants) and mineralization (like corals). The results, published in Nature Communications, have already garnered 30,000 views and 16 citations.
The most impressive part — this isn’t just a lab fantasy. At the 2025 Venice Architecture Biennale, the team 3D-printed a 3.3-meter-tall structure from this material that captures up to 18 kg of CO₂ per year — equivalent to a 20-year-old pine tree.
The Core Idea
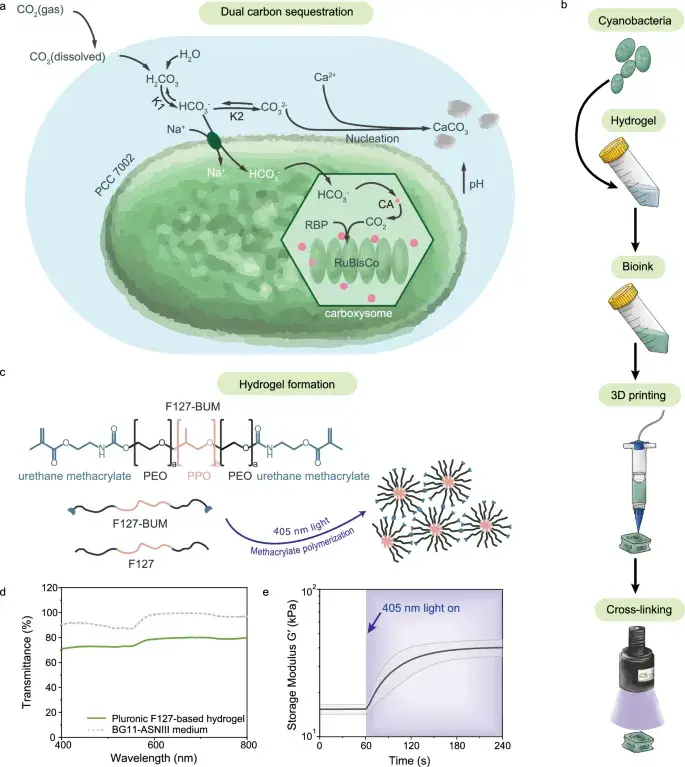
Imagine an aquarium filled with transparent jelly, inside which microscopic green «algae» float. They live, reproduce, absorb CO₂ from the air through photosynthesis, release oxygen — and meanwhile force calcium from the surrounding environment to turn into stone. Literally: living organisms inside the gel build themselves a mineral scaffold, and in the process make the entire material stronger.
Cyanobacteria are the oldest photosynthetic organisms on Earth (over 2.5 billion years old). They’re the ones that originally oxygenated Earth’s atmosphere. Unlike plants, they’re bacteria — prokaryotes without a nucleus. The strain Synechococcus sp. PCC 7002 used in this work is a marine cyanobacterium capable of efficient photosynthesis even under low light conditions.
Hydrogel is a three-dimensional polymer network that retains large amounts of water (up to 90% or more by mass). Think of gummy bears or contact lenses — those are hydrogels. In this work, the Pluronic F127-based hydrogel is transparent, 3D-printable, and transmits light for photosynthesis.
The key innovation is a dual carbon capture mechanism:
- Photosynthesis: cyanobacteria absorb CO₂ and convert it into biomass (organic carbon)
- Biomineralization (MICP): bacteria raise the pH of their environment, causing calcium carbonate to precipitate — essentially forming limestone right inside the gel
The second mechanism is critically important. Biomass is unstable: when an organism dies, its carbon returns to the atmosphere. But mineral carbonate is geological storage. Carbon locked as CaCO₃ stays sequestered for millennia, just like in rock formations.
MICP (Microbially Induced Carbonate Precipitation) is a process where microorganisms create conditions for carbonate mineral deposition. Conventional MICP uses ureolytic bacteria that require urea and produce toxic ammonia. Photosynthetic MICP is an environmentally clean alternative: the only «byproduct» is oxygen.

Fig. 1: Engineering photosynthetic living materials. (a) Dual CO₂ capture pathway — photosynthesis + mineralization. (b) Chemical structure of F127-BUM hydrogel and photo-crosslinking. (c-d) Optical and mechanical properties. Source: Dranseike et al., Nature Communications, 2025
How It Works
The «Living Ink» Recipe
The material’s backbone is a modified block copolymer Pluronic F127, familiar to pharmacists as Poloxamer 407. It’s functionalized with bis-urethane methacrylate groups (yielding F127-BUM), enabling UV-triggered structural fixation. The final bioink formulation:
- 13.2 wt% F127 + 7.3 wt% F127-BUM + 0.1 wt% LAP photoinitiator
- Synechococcus sp. PCC 7002 cyanobacteria at optical density OD₇₃₀ = 0.8
- BG11-ASNIII nutrient medium (simulated seawater)
- Calcium chloride (5 mM) added from day 5 to initiate mineralization
The key property: the gel is thermoresponsive — viscous enough for printing at room temperature, then irreversibly crosslinked after UV exposure (405 nm, 60 seconds). It remains transparent, transmitting 76 ± 3% of visible light.
Two 3D Printing Methods
The authors demonstrated two approaches:
Direct Ink Writing (DIW) — classic extrusion-based 3D printing. Bioink is extruded through a 0.41 mm nozzle at 40–60 kPa pressure. Suitable for disc samples and simple structures.
Volumetric printing (Tomolite) — this borders on science fiction. Instead of layer-by-layer printing, it uses computed axial lithography: a computer projects a series of 2D images into a rotating volume of bioink, and the entire 3D structure forms simultaneously. A single print creates a lattice 22 mm tall with 960 pores (0.5–1.0 mm diameter) for optimal light and nutrient penetration.
Computed Axial Lithography (CAL) is a revolutionary 3D printing method where an object forms all at once within a volume of photopolymer, in seconds, without layer-by-layer construction. The principle is like computed tomography in reverse: instead of scanning an object with X-rays, a projector creates an object with light.
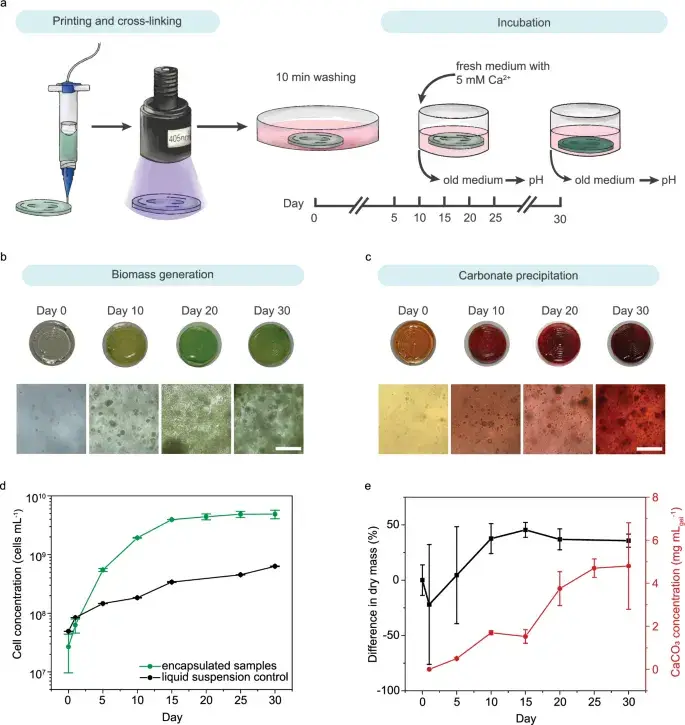
Fig. 2: Dual carbon sequestration. Experimental timeline, cell growth visualization, Alizarin Red S staining of carbonate deposits, biomass growth kinetics over 30 days. Source: Dranseike et al., Nature Communications, 2025
Bacterial Living Conditions
After printing, constructs are placed in a controlled environment:
- Temperature: ~25°C
- Illumination: 180 µmol photons m⁻² s⁻¹ (full-spectrum white light)
- Photoperiod: 12 hours light / 12 hours dark
- Medium replacement: every 5 days
- From day 5: addition of 5 mM CaCl₂ to trigger mineralization
Results
CO₂ Capture: The Numbers
The central question: how much carbon dioxide does the material actually capture?
| Period | CO₂ capture per gram of hydrogel |
|---|---|
| 30 days | 2.2 ± 0.9 mg |
| 400 days | 26 ± 7 mg |
Over 30 days, 4.8 ± 2 mg CaCO₃ precipitates per milliliter of gel. Over 400 days, the majority of captured carbon transitions into mineral form — geologically stable storage.
For comparison: recycled concrete captures roughly 6.7 mg CO₂ per gram through chemical mineralization. The ETH Zurich living material captures 26 mg/g over 400 days — nearly 4 times more. And it keeps going.
Growth and Strengthening
The bacteria don’t just survive — they make the material stronger:
- Final cell concentration: 5 × 10⁹ cells per mL of gel by day 30
- Storage modulus (G’) grows from 5.4 kPa (day 2) to 10.1 kPa (day 30) — bacteria nearly double the stiffness
- 3D-printed lattices at 400 days: modulus of 111 kPa — 10 times stronger than initial values
The material literally grows and petrifies — biomass and minerals comprise ~45% of the final sample mass by day 30.
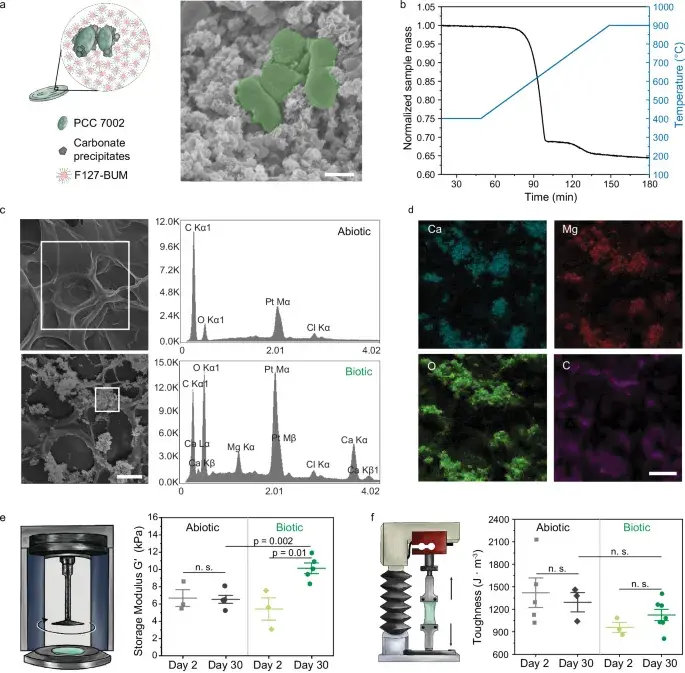
Fig. 3: Carbonate formation. SEM images of mineral deposits around cells, thermogravimetric analysis, elemental mapping (Ca, Mg, O, C), and mechanical property dynamics over time. Source: Dranseike et al., Nature Communications, 2025
Mineral Composition
Electron microscopy and X-ray diffractometry revealed that the precipitated minerals are magnesium-substituted calcite (Ca₇₈Mg₂₂)CO₃. The calcium-to-magnesium ratio is 78 ± 5% to 22 ± 5%. In essence, the bacteria build the same mineral inside the gel that forms coral reefs and limestone cliffs.
Venice Biennale: From Test Tube to Architecture
The most spectacular demonstration was the Picoplanktonics project at the Canada Pavilion of the 2025 Venice Architecture Biennale. Using «living ink» (sand + biocompatible binder + cyanobacteria), the team robotically 3D-printed tree trunk-shaped structures. The largest stood 3.3 meters tall.
Each structure captures up to 18 kg CO₂ per year — equivalent to a 20-year-old pine tree in a temperate climate. To sustain the bacteria, the pavilion maintained controlled conditions (light, humidity, warmth), and a team of «caretakers» tended to the living structures throughout the exhibition (May–November 2025).

Fig. 4: 3D-printed living structures. Lattice design for optimal light penetration, structural evolution over 365 days, mineral scaffold after thermal decomposition of organics, coral-inspired textured surface. Source: Dranseike et al., Nature Communications, 2025
Critical Assessment
This work was published in Nature Communications — a peer-reviewed journal with an impact factor of 16.6. This is not a preprint. Nevertheless, every scientific result should be examined critically.
Strengths
-
Dual mechanism — a fundamental departure from all previous biological CO₂ capture approaches. Mineralization provides long-term carbon storage, not just temporary biomass binding.
-
Full-scale demonstration — from laboratory discs to a 3.3-meter architectural structure at one of the world’s premier exhibitions. This is not theoretical work; it’s a functioning technology.
-
Environmental cleanliness — unlike ureolytic MICP, no urea is needed and no toxic ammonia is produced. The only byproduct is oxygen.
Limitations
-
Modest capture rate. 2.2 mg CO₂ per gram per month is orders of magnitude less than industrial direct air capture (DAC) systems. The authors openly acknowledge: replacing chemical technologies is a distant goal.
-
Environmental dependence. Bacteria require light, warmth, humidity, and saltwater. Biomass growth plateaus around day 25 as inner layers receive progressively less light. Transparency drops from 76% to ~30%.
-
Unknown long-term stability. The 400-day experiment showed promising results, but what happens after 10 or 50 years? The authors did not conduct a life cycle assessment (LCA) or model behavior under real atmospheric conditions.
Open Questions
- What is the net carbon balance accounting for hydrogel production, printing, and maintenance?
- Can cyanobacteria be genetically engineered to increase photosynthesis rates without losing MICP capability?
What’s Next
The authors outline several development directions:
- Direct O₂ electrode measurements for precise quantification of photosynthesis
- Computational modeling of pH-dependent CO₂ assimilation
- Genetic engineering of cyanobacteria for enhanced photosynthetic activity
- Optical waveguides within the material structure to deliver light deeper
- Life cycle assessment (LCA) — cradle-to-grave carbon footprint calculation
The technology is still far from replacing concrete. But it points toward an entirely new class of building materials — living, self-strengthening, and actively reducing atmospheric CO₂. As Mark Tibbitt put it: «We see our living material as a low-energy and environmentally friendly approach that can bind CO₂ from the atmosphere and complement existing chemical processes for carbon sequestration.»
And Andrea Shin Ling, who created the Venice Biennale structures, adds: «The bacteria cooperate in a human-initiated fabrication process. A bioprint might dry out if the air is too dry that week, and many bacteria die. But because the system is regenerative, the bacteria population has the potential to restore itself when favorable conditions return.»
Buildings that breathe, grow, and absorb carbon dioxide — it sounds like science fiction. But it was already standing at the Venice Biennale.
Frequently Asked Questions
How does the living material compare to regular concrete in terms of strength?
It doesn’t — yet. The elastic modulus of the living hydrogel (~10–111 kPa) is orders of magnitude lower than concrete (~30 GPa). This isn’t a replacement for load-bearing structures, but rather a material for facade panels, cladding, and decorative elements that simultaneously capture CO₂.
How long do the bacteria survive inside the gel?
In experiments, bacteria remained alive and active for over 400 days. Moreover, the system is regenerative: even if part of the population dies (e.g., during drying), surviving bacteria can restore the colony once favorable conditions return.
Can this material be used in ordinary buildings?
At this stage — only in specially equipped conditions (light, humidity, periodic maintenance). Mass application requires further advances in autonomy and environmental resilience.
How much CO₂ could an entire building made from this material capture?
Based on Venice Biennale data, a single 3.3-meter structure captures ~18 kg CO₂ per year. Covering a 1,000 m² building facade with such material could theoretically capture hundreds of kilograms of CO₂ annually. But precise calculations require a full life cycle assessment.
Why is photosynthetic MICP better than traditional MICP?
Traditional MICP uses ureolytic bacteria that require urea and produce ammonia (NH₃) — a toxic byproduct. Photosynthetic MICP runs on CO₂ and water, releasing only oxygen. This makes it safe for use in residential spaces and open environments.
References
Related
Related Articles
CO₂ in Your Blood: Why Air Could Turn Toxic by 2076
A 20-year study of 70,000+ blood samples shows bicarbonate rising in lockstep with atmospheric CO₂ — set to breach safe limits by 2076.
Bacteria That Eat Tumors: Cancer Therapy
Engineers gave Clostridium sporogenes quorum sensing — bacteria find tumors, wait for backup, then destroy cancer from within.
CO₂ to Fuel: Tungsten Photocatalyst Breakthrough
A tungsten-based photocatalyst converts CO₂ and water into fuel using only sunlight — mimicking photosynthesis at room temperature.