CO₂ to Fuel: Tungsten Photocatalyst Breakthrough
Authors: Yu Huang, Xianjin Shi, Hongna Zhang, Junji Cao, Shuncheng Lee
Why It Matters
Every year, humanity pumps over 37 billion tonnes of carbon dioxide into the atmosphere. At the same time, we burn oil to produce gasoline, jet fuel, and diesel — fuels that planes and ships still can’t do without. What if we could close that loop — take CO₂ from the air, add water and sunlight, and produce the raw material for the same gasoline?
That’s exactly what researchers from the Chinese Academy of Sciences and the Hong Kong University of Science and Technology set out to do. In a paper published in Nature Communications on January 31, 2026, Yu Huang’s team presented a photocatalyst that converts CO₂ and water into carbon monoxide (CO) — a key «building block» for synthesizing liquid fuels. And it does so by copying one of nature’s most elegant tricks.
The Core Idea
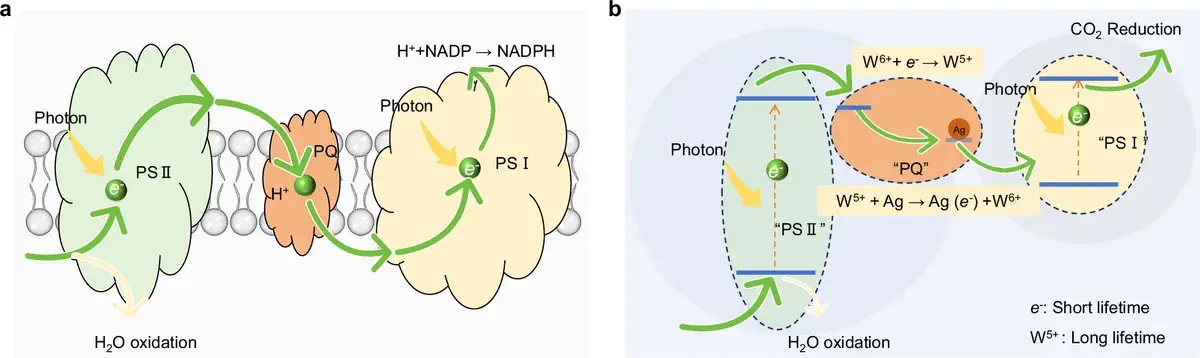
Plants have been converting CO₂ for over three billion years. Inside chloroplasts, light splits water, and the freed electrons travel along a chain of carriers to CO₂ molecules, turning them into organic matter. One critical link in this chain is plastoquinone — a molecule that acts as an electron «staging area»: it accumulates electrons when light is abundant and releases them when needed.
Plastoquinone — an electron carrier molecule in plant chloroplasts. It accepts electrons from Photosystem II and passes them along the chain. It can temporarily «store» charge, smoothing the mismatch between the speed of light absorption and the pace of chemical reactions.
The authors of this study replicated that idea in an inorganic material. Their «plastoquinone» is tungsten trioxide (WO₃) modified with silver nanoparticles. Under illumination, tungsten atoms shift from a W⁶⁺ state to W⁵⁺, storing electrons, and then release them back. The result is a kind of «charge battery» — a charge reservoir, as the authors call it.
Tungsten trioxide (WO₃) — a semiconductor oxide that absorbs visible light (up to ~480 nm). It is known for its ability to switch the valence state of tungsten (W⁶⁺ ↔ W⁵⁺), which makes it suitable as an «electron buffer.»
How It Works
The system comprises three components, each with a distinct role:
Ag/WO₃ — the charge reservoir. It absorbs light and accumulates electrons through W⁶⁺ → W⁵⁺ transitions. Silver nanoparticles enhance charge separation through the plasmonic effect.
CoPc (cobalt phthalocyanine) — the catalytic center. A molecular catalyst that binds directly to CO₂ and reduces it to CO. Electrons from the «reservoir» flow to CoPc, scavenging photogenerated holes and maintaining a high electron density at the active sites.
Water — the electron source. Unlike most comparable systems, no organic «sacrificial» reagents such as triethanolamine (TEOA) are consumed in the process. Water is oxidized instead, donating electrons.
Sacrificial reagent — a substance that is irreversibly oxidized to supply electrons for CO₂ reduction. Most photocatalytic systems rely on such additives. Operating without them is a significant achievement that brings the technology closer to practical viability.
To use a factory analogy: WO₃ is the battery station, silver is the transformer substation, CoPc is the assembly floor, and water is the free raw material delivered in unlimited supply.
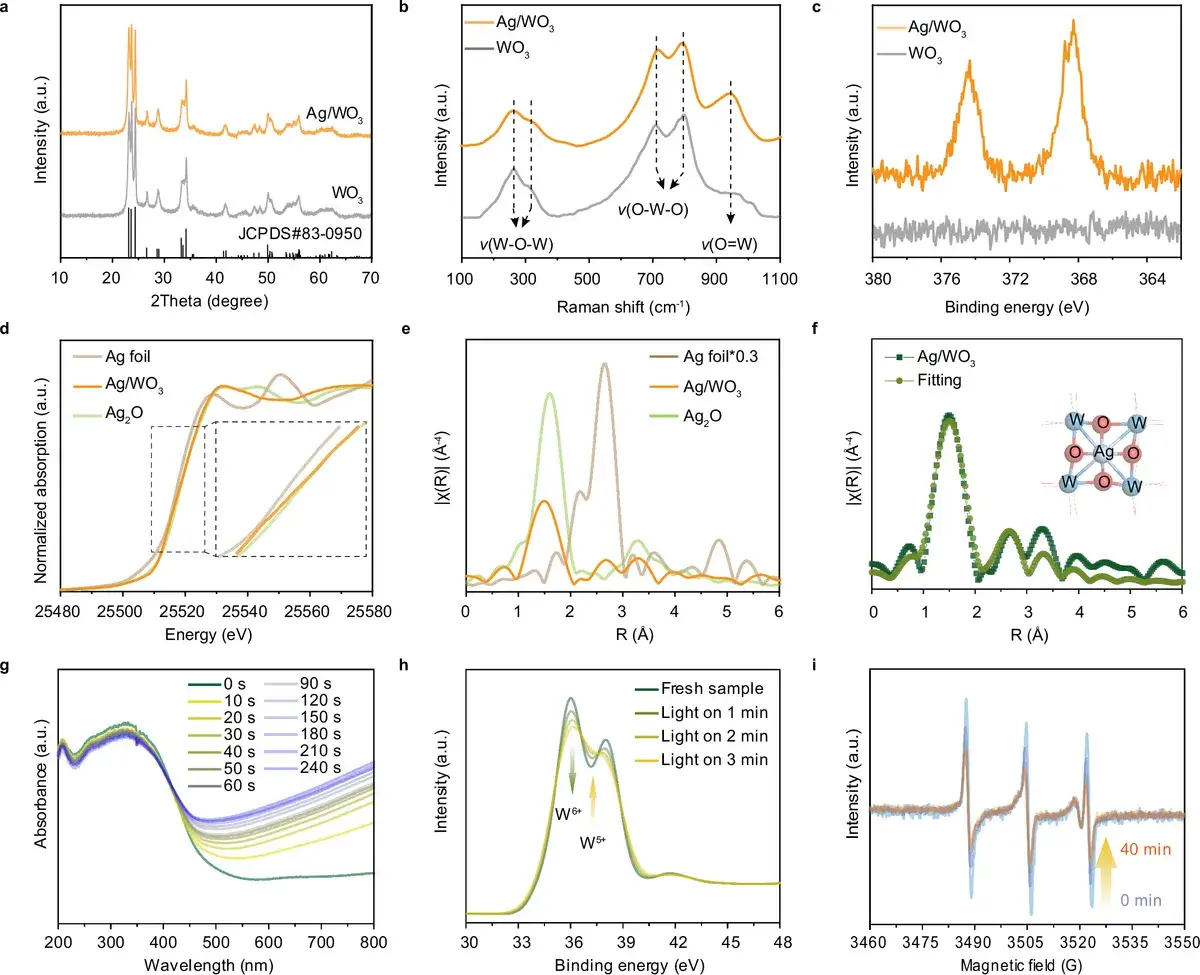
Fig. 2: Catalyst characterization. X-ray diffraction (XRD), Raman spectroscopy, X-ray photoelectron spectroscopy (XPS), and XANES confirm successful formation of the CoPc/Ag/WO₃ heterostructure and W⁶⁺ → W⁵⁺ transitions. Source: Huang et al., Nature Communications, 2026
Results
The CoPc/Ag/WO₃ catalyst achieved a CO production rate of approximately 1.5 mmol per gram of CoPc per hour — a 100-fold increase over bare cobalt phthalocyanine without any support.
For perspective: typical CoPc heterostructures produce 0.1–0.3 mmol/g/h. A close competitor — a CoPc system on a covalent triazine framework (CTF) published in the same Nature Communications in 2024 — achieved ~0.46 mmol/g/h. The present work shows roughly a threefold advantage.
An important detail: the authors claim universality of the approach. Ag/WO₃ boosted the performance not only of CoPc but also of other active components, suggesting that the «charge reservoir» concept itself works rather than being a lucky one-off pairing.
| System | CO, mmol/g/h | Sacrificial reagent |
|---|---|---|
| CoPc/Ag/WO₃ (this work) | ~1.5* | No (H₂O) |
| CoPc/CTF (Nat. Commun. 2024) | ~0.46 | No (KHCO₃) |
| Co-COF systems | 1.6–16.4 | Yes (TEOA) |
| N-Cu₂O/quartz (arXiv 2025) | ~0.17 | No (H₂O) |
*Normalized to CoPc mass, not total catalyst mass — see Critical Assessment below.
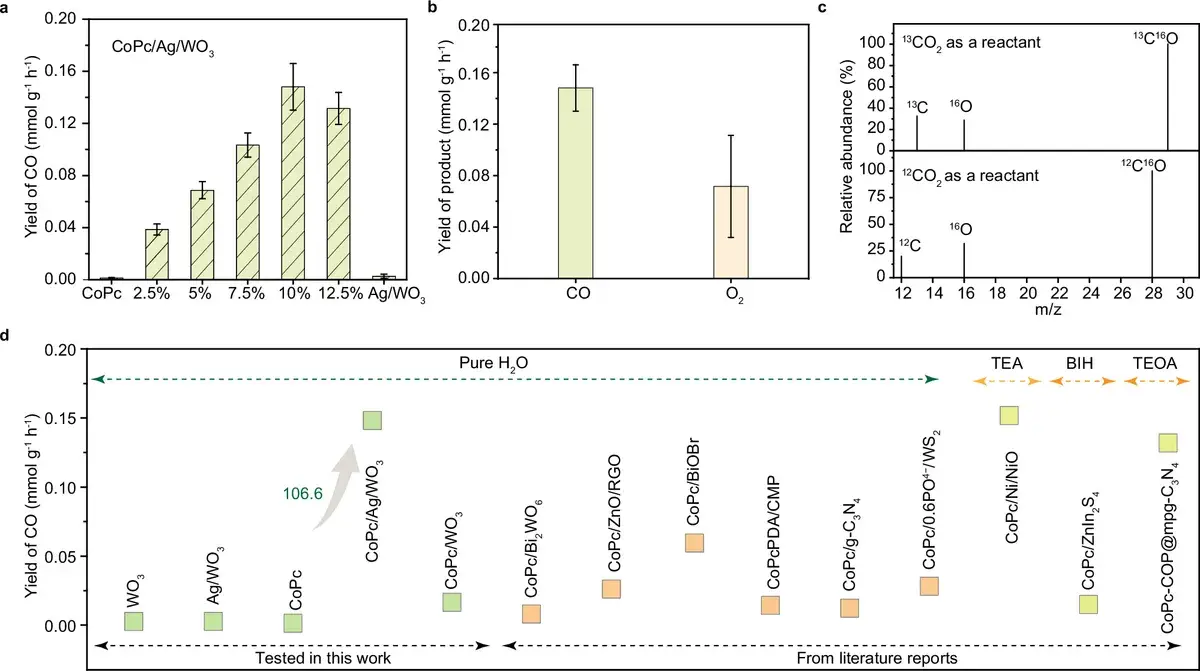
Fig. 3: Results. CO generation kinetics, carbon origin verification via ¹³CO₂ isotope labeling, and comparative chart with other photocatalytic systems. Source: Huang et al., Nature Communications, 2026
Critical Assessment
The work was published in the peer-reviewed journal Nature Communications (reviewer: Mingwu Tan and anonymous experts). Nevertheless, several points deserve careful consideration.
Strengths:
- Operating without a sacrificial reagent is rare in a field where 95% of systems depend on organic additives
- An original bioinspired concept with a clear functional separation between components
- Demonstrated universality of the approach across multiple active components
Limitations:
- Performance is normalized to CoPc mass, not total catalyst mass. If CoPc constitutes, say, 5% of the composite, the real productivity per gram of catalyst would be ~0.075 mmol/g/h — a far more modest figure
- WO₃ absorbs light only up to ~480 nm (band gap ~2.6–2.8 eV). That covers just 6–8% of the solar spectrum, making real-world solar efficiency extremely low
- Apparent quantum efficiency (AQE) is not highlighted as a headline figure. For systems without sacrificial reagents, it is typically well below 1%
- Long-term stability data are not emphasized in the paper. Silver is prone to oxidation and aggregation; CoPc tends to deactivate over time
Open questions:
- Has oxygen evolution (O₂) been confirmed in stoichiometric ratio with CO? Without this, one cannot rule out that electrons come from surface contaminants rather than water
- What is the actual contribution of silver’s plasmonic effect versus the tungsten valence oscillation mechanism?
Important note: the plastoquinone analogy is elegant but has limits. Plastoquinone is a mobile molecule that physically shuttles between protein complexes in the thylakoid membrane. Tungsten atoms in the WO₃ crystal lattice don’t move. The parallel works at the functional level (temporary charge storage) but not at the mechanistic level.
It is also worth noting a systemic challenge across the entire field of photocatalytic CO₂ reduction: without isotopic labeling using ¹³CO₂, it is impossible to definitively prove that the produced CO originates from carbon dioxide rather than from organic contaminants on the catalyst surface.
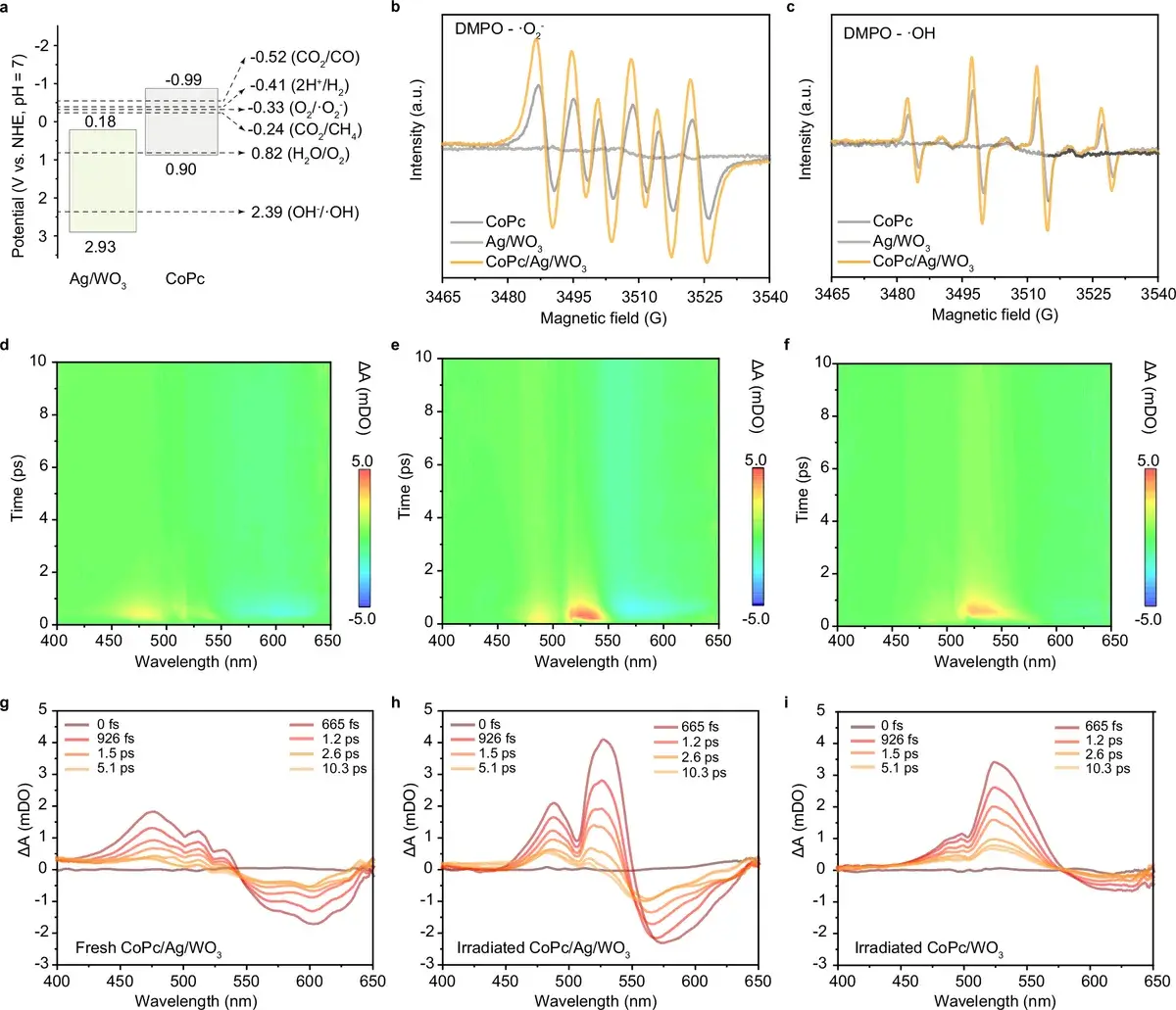
Fig. 4: Mechanism. Band diagram, ESR spectra, and transient absorption spectroscopy data reveal the role of tungsten valence oscillation (W⁶⁺ ↔ W⁵⁺) in electron accumulation and transfer. Source: Huang et al., Nature Communications, 2026
What’s Next
Carbon monoxide (CO), which this catalyst produces, is not a fuel in itself. It is an intermediate — a «building block» from which gasoline, jet fuel, and diesel can be produced using well-established industrial processes.
Two main routes exist:
- Fischer-Tropsch synthesis: CO + H₂ → liquid hydrocarbons. The technology has been known since the 1920s
- Via methanol: CO → methanol → gasoline (ExxonMobil’s MTG process). Already operating at industrial scale
China is actively pursuing both pathways. The Dalian Institute of Chemical Physics (DICP, CAS) has launched a demonstration plant producing 1,000 tonnes of gasoline per year from CO₂ — though using thermocatalysis (Na-Fe₃O₄/HZSM-5) at high temperatures and pressures.
Meanwhile, Swiss company Synhelion commissioned its DAWN plant in Germany in 2024, producing synthetic fuel from solar heat at 1,500°C. Its first commercial plant, RISE, with a capacity of 1,000 tonnes per year, is planned for Spain by 2027.
Huang and colleagues take a different approach: photocatalytic, at ambient conditions, without extreme temperatures. Industrial scale is still far off — challenges of stability, quantum yield, and scaling remain. But the concept of a tungsten «charge battery» that mirrors photosynthesis logic opens a compelling direction for future research.
The full «sun-to-fuel» chain currently achieves just 13–20% efficiency even in the best thermochemical systems. Photocatalysis lags further behind. But this is how science progresses — from an elegant principle to a working technology, one step at a time.
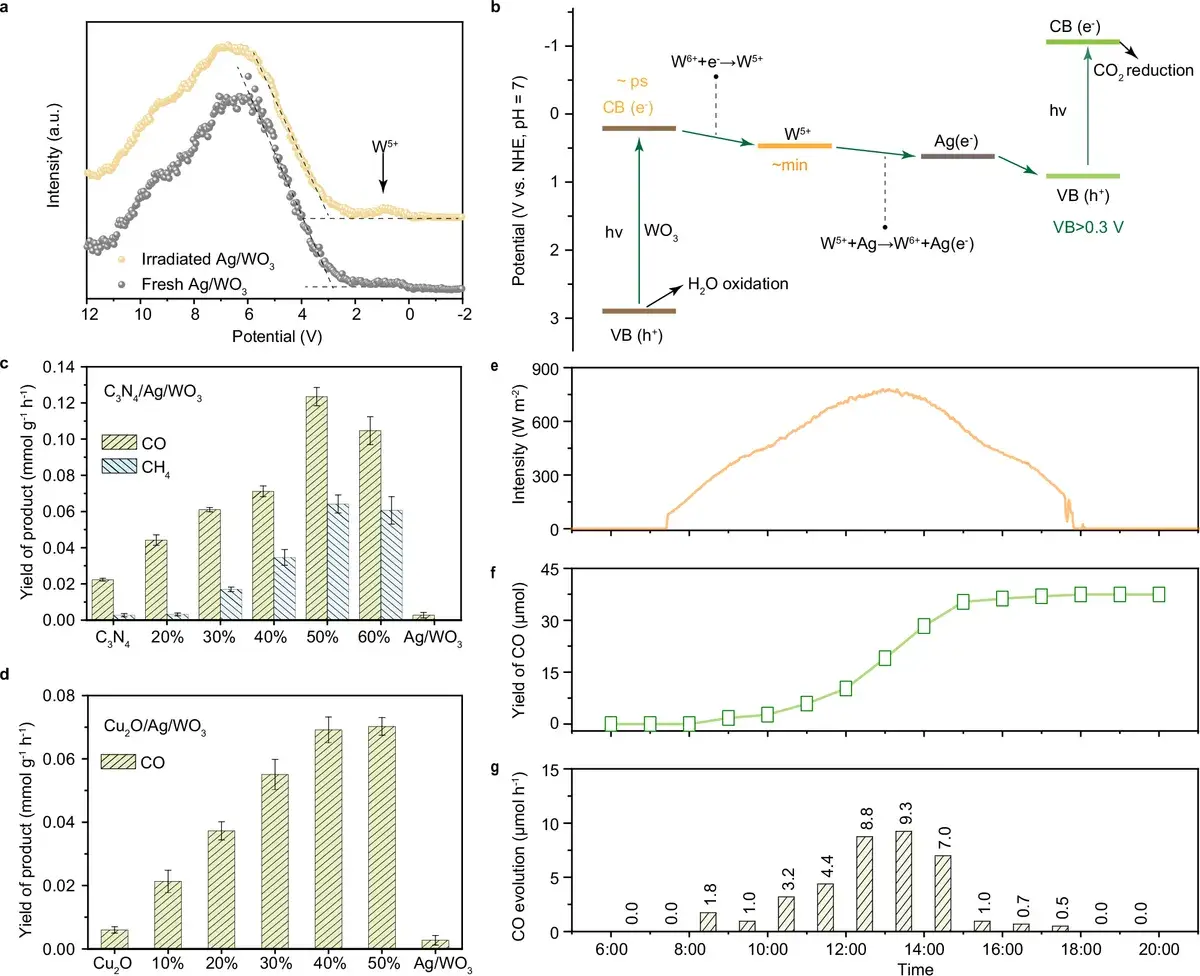
Fig. 5: Universality and practical tests. The charge reservoir mechanism works with various catalytic centers (not just CoPc). Outdoor tests confirm the system operates under real sunlight. Source: Huang et al., Nature Communications, 2026
Frequently Asked Questions
Can this catalyst replace oil refineries?
No — this is fundamental research at this stage. The catalyst produces CO, which is only an intermediate product that must then be processed into fuel. Current scales involve milligrams of catalyst in a laboratory reactor. Years of work on stability, efficiency, and scale-up will be required before any industrial application.
How is this different from solar panels plus electrolysis?
Solar panels generate electricity that can split water into hydrogen, which is then combined with CO₂ to synthesize fuel. That’s a two-step process. A photocatalyst does everything in a single step — light directly drives the chemical reaction. The advantage is simplicity; the drawback is much lower efficiency so far.
Why does it matter to work without sacrificial reagents?
Sacrificial reagents are organic substances that are irreversibly consumed in the reaction, donating electrons in place of water. They make experiments easier but strip the technology of practical meaning: if producing «green» fuel requires burning other organics, the carbon cycle doesn’t close.
What is a fuel «building block»?
CO (carbon monoxide) is a molecule consisting of one carbon atom and one oxygen atom. By itself, it is toxic and not a fuel. But when mixed with hydrogen, CO becomes syngas — a feedstock from which gasoline, jet fuel, and diesel molecules can be assembled via Fischer-Tropsch synthesis or through methanol conversion.
Is this truly «artificial photosynthesis»?
Partly. The system replicates one specific element of photosynthesis — temporary electron storage, as plastoquinone does. But real photosynthesis involves dozens of coordinated reactions with hundreds of proteins. This catalyst performs only the first step: CO₂ → CO. A complete «artificial leaf» that produces ready-made fuel from air and water remains a distant goal.
References
Related
- Sustainable all-weather CO2 utilization by mimicking natural photosynthesis in a single material
- Integrable utilization of intermittent sunlight and residual heat for on-demand CO2 conversion with water
- Molecular catalyst coordinatively bonded to organic semiconductors for selective light-driven CO2 reduction in water
Related Articles
China Builds the First Reactor That Eats Nuclear Waste
The world's first megawatt-scale ADS reactor launches in Huizhou, 2027 — designed to shrink nuclear waste danger from 100,000 years to under 300.
CO₂ in Your Blood: Why Air Could Turn Toxic by 2076
A 20-year study of 70,000+ blood samples shows bicarbonate rising in lockstep with atmospheric CO₂ — set to breach safe limits by 2076.
AI Finds 25 Rare-Earth-Free Magnets in 67,000
AI-curated database of 67,000 magnetic materials reveals 25 high-temperature alternatives to rare-earth magnets for EVs.