CAR-T Immune Reset Cured 3 Autoimmune Diseases
Authors: Fabian Müller, Georg Schett, Andreas Mackensen
A 47-year-old woman in Erlangen, Germany, had three autoimmune diseases attacking her simultaneously. Her own immune system was destroying her red blood cells, her platelets, and forming clots in her vessels. She had cycled through every standard therapy. Nothing held. Then her doctors extracted a vial of her T cells, spent a few weeks engineering them in a lab, and infused them back. Twenty-five days later, her blood counts were normal. Fourteen months on — still normal, still off all medications.
That case report, published in Med in early 2026, is dramatic but not isolated. It sits alongside a formal clinical trial that tested the same approach in 24 people across three different autoimmune diseases. The results have just landed in Nature Medicine, and they mark the strongest evidence yet that a single infusion of reprogrammed immune cells can reboot a haywire immune system.
The Weapon Borrowed From Cancer
CAR-T cells (Chimeric Antigen Receptor T cells) are a patient’s own T cells, extracted and genetically engineered to carry a synthetic receptor that recognizes a specific target on other cells. Originally developed to hunt cancer cells, they are now being repurposed against autoimmune disease.
The therapy is called CAR-T — chimeric antigen receptor T cell therapy. It was built to fight blood cancers like leukemia, where it has been saving lives since 2017. The idea is blunt and effective: take a patient’s T cells out, bolt on a synthetic receptor that locks onto a surface marker called CD19, multiply them, and put them back. These engineered cells then hunt every B cell in the body that carries CD19 on its surface.
In cancer, the target is malignant B cells. In autoimmune disease, the target is the B cells that produce autoantibodies — the rogue molecules that attack the body’s own tissues. Wipe them out, and the source of the attack disappears.
But here is the part that makes the approach more than just aggressive immunosuppression. After the engineered T cells clear the slate, new B cells eventually grow back from precursors in the bone marrow. These fresh B cells are naive. They carry no memory of how to attack the body. The immune system restarts, and the autoimmune «program» is gone.
CD19 is a protein found on the surface of nearly all B cells, from early precursors to mature cells and short-lived antibody-secreting plasmablasts. Critically, long-lived plasma cells in the bone marrow do not express CD19, which means protective antibodies from past vaccinations are largely preserved after treatment.
The Erlangen Experiment, Four Years Running
The research group behind this work is based at the University Hospital Erlangen in Germany, led by Georg Schett and Andreas Mackensen. They didn’t start with a 24-patient trial. They started in 2021 with a single lupus patient who had run out of options.
That first patient went into drug-free remission. Then four more. A 2022 paper in Nature Medicine reported the initial five, all with systemic lupus erythematosus, all in sustained remission. A 2024 case series in the New England Journal of Medicine expanded the cohort and added patients with systemic sclerosis and inflammatory myopathy. The results held.
The CASTLE trial — short for «CAR-T cells in Systemic B cell mediated Autoimmune disease» — is the formal phase ½ trial that grew from those early cases. It used a product called zorpocabtagene autoleucel (Zorpo-cel), manufactured by Miltenyi Biomedicine, with a 4-1BB co-stimulatory domain identical to the one used in tisagenlecleucel (Kymriah), the first CAR-T approved for cancer.
24 Patients, Three Diseases, Six Months Off Every Drug
The trial enrolled 24 patients with severe, treatment-refractory disease across three baskets:
Ten had systemic lupus erythematosus (SLE) — a disease where antibodies attack the skin, joints, kidneys, and brain. Nine had systemic sclerosis (SSc) — a condition that hardens the skin and scars the lungs. Five had idiopathic inflammatory myopathies (IIM) — diseases that destroy muscle tissue. The median patient had failed four prior immunosuppressive regimens. Their median disease duration was four years.
Each patient received lymphodepletion chemotherapy (cyclophosphamide plus fludarabine — the same conditioning used before CAR-T in cancer), followed by a single infusion of Zorpo-cel.
Lymphodepletion is a short course of chemotherapy given before the CAR-T infusion. It temporarily suppresses the patient’s existing immune cells, creating space for the engineered T cells to expand and do their work.
The results at 24 weeks: 22 of 24 patients (92%) met the predefined efficacy endpoints for their specific disease. Nine of ten lupus patients achieved DORIS remission. All nine sclerosis patients showed no disease progression. Four of five myopathy patients reached a major or moderate response by ACR/EULAR criteria.
Every single patient — all 24 — was completely free of glucocorticoids and all immunosuppressive medications for the entire six-month observation period. For patients who had been on multiple drugs for years, that fact alone is unprecedented.
What Happened Inside the Body
The engineered T cells peaked at a median of 140 cells per microliter of blood on day 10 after infusion. B cells vanished within seven days — complete depletion. They stayed gone for a median of 83 days.
When B cells eventually returned, their composition had changed. Almost all were naive B cells. Memory B cells and plasmablasts — the subtypes responsible for producing autoantibodies — were virtually absent. In lupus patients, anti-dsDNA antibodies (a hallmark of the disease) seroconverted to negative within six months.
This is the biological signature of the immune reset. The old, misprogrammed B cell repertoire is erased. The new one grows without carrying forward the autoimmune instructions.
Where It Breaks Down
The CASTLE trial was published in Nature Medicine and has undergone peer review, though it remains a phase ½ study with inherent limitations.
Twenty-four patients is a small number, and all were treated at a single center in Erlangen. The follow-up period was six months — long enough to demonstrate feasibility, too short to know whether remission lasts years. The Erlangen group’s earlier patients from 2021-2022 are still in remission at three-plus years, which is encouraging, but those were not part of a formal controlled trial.
Two patients did not meet their efficacy endpoints. The published abstract does not detail why, but the most likely explanations involve irreversible organ damage that preceded treatment — fibrosis in the lungs or kidneys that no immune reset can undo. This underscores a critical point: CAR-T works by stopping the autoimmune attack, not by repairing damage already done. Timing matters enormously.
One patient experienced the trial’s only dose-limiting toxicity: cytomegalovirus (CMV) reactivation leading to pneumonitis and kidney damage. Deep B cell depletion leaves patients temporarily vulnerable to viral infections, and CMV is a known risk during immunosuppression. No patient developed cytokine release syndrome above grade 2 (mild, self-resolving), and no cases of neurotoxicity occurred — a substantially better safety profile than CAR-T in cancer, where these side effects are frequent and sometimes severe.
Then there is cost. In oncology, a single CAR-T infusion runs between $373,000 and $530,000 in the United States. Autoimmune pricing is not yet established, but estimates hover around EUR 360,000 in Europe. For diseases like lupus that affect roughly 5 million people worldwide, or rheumatoid arthritis at over 17 million, current manufacturing capacity could not scale to meet demand even at unlimited budgets. Miltenyi’s point-of-care manufacturing model — producing the cells at the hospital rather than shipping them to a central facility — may eventually lower costs, but this remains aspirational.
The Road From Single Center to Standard of Care
The Erlangen group is not alone anymore. A companion paper in the same issue of Nature Medicine reports results from eight children with autoimmune diseases treated with the same approach — four with lupus, three with dermatomyositis, one with sclerosis — with similar efficacy and safety outcomes. Research groups in the Netherlands, Italy, and the United States are launching their own trials.
The next step is a pivotal phase 3 study, which will need to be multicenter, larger, and include a control arm — likely patients receiving standard immunosuppressive therapy. If those results hold, regulatory approval for autoimmune indications could follow within three to five years.
Meanwhile, researchers are exploring in vivo CAR-T delivery, where the genetic cargo is delivered directly to T cells inside the patient’s body using lipid nanoparticles, eliminating the need for cell extraction, engineering, and reinfusion. If that technology matures, it could collapse both cost and manufacturing bottlenecks.
«The major breakthrough in this entire endeavor is that, for the very first time in severe autoimmune diseases, you actually have a treatment-free period, ” Fabian Müller, the trial’s lead author, told Medscape. For patients who have spent years cycling through immunosuppressive drugs — each with its own side effects, none truly curative — that sentence carries weight.
Frequently Asked Questions
Why don’t the new B cells attack the body again after the reset?
After CAR-T cells destroy all CD19-positive B cells, the body regenerates its B cell pool from bone marrow precursors. These new B cells are immunologically naive — they have not been exposed to the self-antigens that triggered the original autoimmune response. In the CASTLE trial, memory B cells and autoantibody-producing plasmablasts were virtually absent when B cells returned after a median of 83 days. The autoimmune «memory» appears to be lost with the old B cell repertoire. Whether this reset is permanent remains an open question, but the earliest patients treated in Erlangen (2021) remain in remission more than three years later.
Does the treatment wipe out immunity from past vaccinations?
Partially, but less than you might expect. CD19 is found on B cells and plasmablasts, but not on long-lived plasma cells that reside in the bone marrow. These plasma cells are the main source of long-term protective antibodies from past vaccinations and infections. Because they lack CD19, they survive the CAR-T treatment. Patients may need booster shots for some vaccines after B cell reconstitution, but they do not lose all acquired immunity.
How much does CAR-T therapy for autoimmune diseases cost?
Currently, there is no approved commercial CAR-T product for autoimmune indications. In oncology, CAR-T therapies cost between $373,000 and $530,000 per infusion in the US, and approximately EUR 360,000 in Europe. The CASTLE trial used Miltenyi’s point-of-care manufacturing platform, which produces the cells at the hospital rather than at a centralized facility, potentially reducing costs. Researchers are also exploring in vivo CAR-T delivery (injecting the genetic material directly) as a future approach that could dramatically lower the price.
Which autoimmune diseases could CAR-T therapy treat beyond lupus, sclerosis, and myopathy?
The CASTLE trial tested three diseases, but the underlying mechanism — eliminating pathogenic B cells and resetting humoral immunity — is relevant to any B cell-mediated autoimmune condition. Early case reports and small trials are exploring CAR-T in multiple sclerosis, myasthenia gravis, rheumatoid arthritis, and autoimmune hemolytic anemia. The companion case report in Med demonstrated remission in a patient with three simultaneous autoimmune conditions (AIHA, ITP, and antiphospholipid syndrome), suggesting broad applicability. Formal trials for each disease will need to establish safety and efficacy individually.
Why did two patients not respond to the treatment?
The published CASTLE data reports that 22 of 24 patients met efficacy endpoints, but does not detail the two non-responders. The most likely explanation is irreversible organ damage — fibrosis in lungs (systemic sclerosis) or kidneys (lupus nephritis) — that preceded treatment. CAR-T eliminates the ongoing autoimmune attack but cannot reverse structural damage already done. This highlights the importance of treating earlier in the disease course, before permanent organ injury accumulates. The Erlangen group has suggested that earlier intervention could improve response rates further.
References
Related
- Anti-CD19 CAR T cell therapy for refractory systemic lupus erythematosus (Nature Medicine, 2022)
- CD19 CAR T-Cell Therapy in Autoimmune Disease — A Case Series with Follow-up (NEJM, 2024)
- Anti-CD19 CAR T cells for pediatric patients with treatment-refractory autoimmune diseases (Nature Medicine, 2026)
- CD19 CAR-T therapy induces remission in refractory AIHA with ITP and antiphospholipid syndrome (Med, 2026)
Context
Related Articles
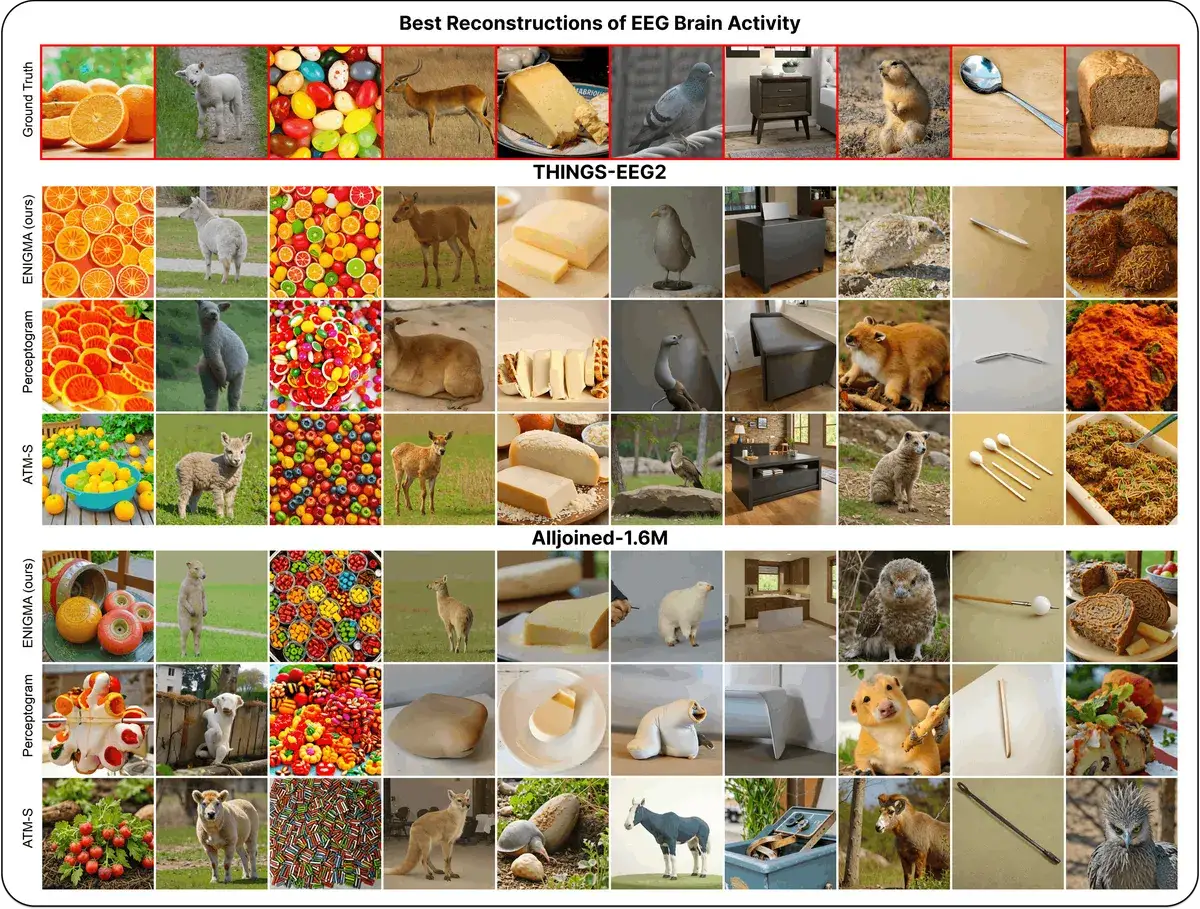
ENIGMA: Reading Minds in 15 Min via EEG
ENIGMA reconstructs images from EEG signals after 15 min of calibration, using under 1% of previous methods' parameters.
Sleep Loss Turns Gut Against Your Brain
Gut bacteria from sleep-deprived mice triggered Alzheimer's-like tau damage in healthy brains. Scientists traced the full molecular chain.
Bacteria That Eat Tumors: Cancer Therapy
Engineers gave Clostridium sporogenes quorum sensing — bacteria find tumors, wait for backup, then destroy cancer from within.