Your Brain Literally Naps When You're Bored: EEG Proof
Authors: Elaine Pinggal, James Jackson, Anikó Kusztor, David Chapman, Jennifer Windt, Sean P.A. Drummond, Tim J. Silk, Mark A. Bellgrove, Thomas Andrillon
You’re in a meeting. The slides are familiar, the speaker’s voice fades into background noise. Your eyes stay open. You nod. But inside your skull, patches of cortex have already clocked out — generating the same electrical waves your brain produces during deep sleep.
A team of neuroscientists at Monash University strapped 64-channel EEG caps onto 63 adults and gave them the most boring task imaginable. What they found upends a basic assumption about wakefulness: your brain doesn’t stay uniformly awake. During monotony, entire cortical regions flicker into microsleep while the rest keeps working. And in people with ADHD, this flickering is dramatically more intense.
The Experiment That Weaponized Boredom
Slow waves — low-frequency oscillations (0.5–4 Hz) in brain electrical activity, typically seen only during deep sleep. Detecting them in an awake person means some neurons have temporarily gone silent.
Thomas Andrillon’s team needed participants who were awake but bored out of their minds. They chose the Sustained Attention to Response Task (SART): digits from 1 to 9 flash on screen, and you press a button for every digit — except 3. Dead simple. That’s the point. The task is so tedious that your brain slips into autopilot within minutes.
Every 45 seconds, the experiment paused to ask: what were you just thinking about? Options ranged from «focused on the task» to «mind wandering» to «mind blanking» — that eerie state where your head feels completely empty. Meanwhile, the EEG recorded every electrical whisper from the cortex.
The study enrolled 32 adults diagnosed with ADHD and 31 without. All were between 18 and 48 years old.
When Cortical Regions Clock Out
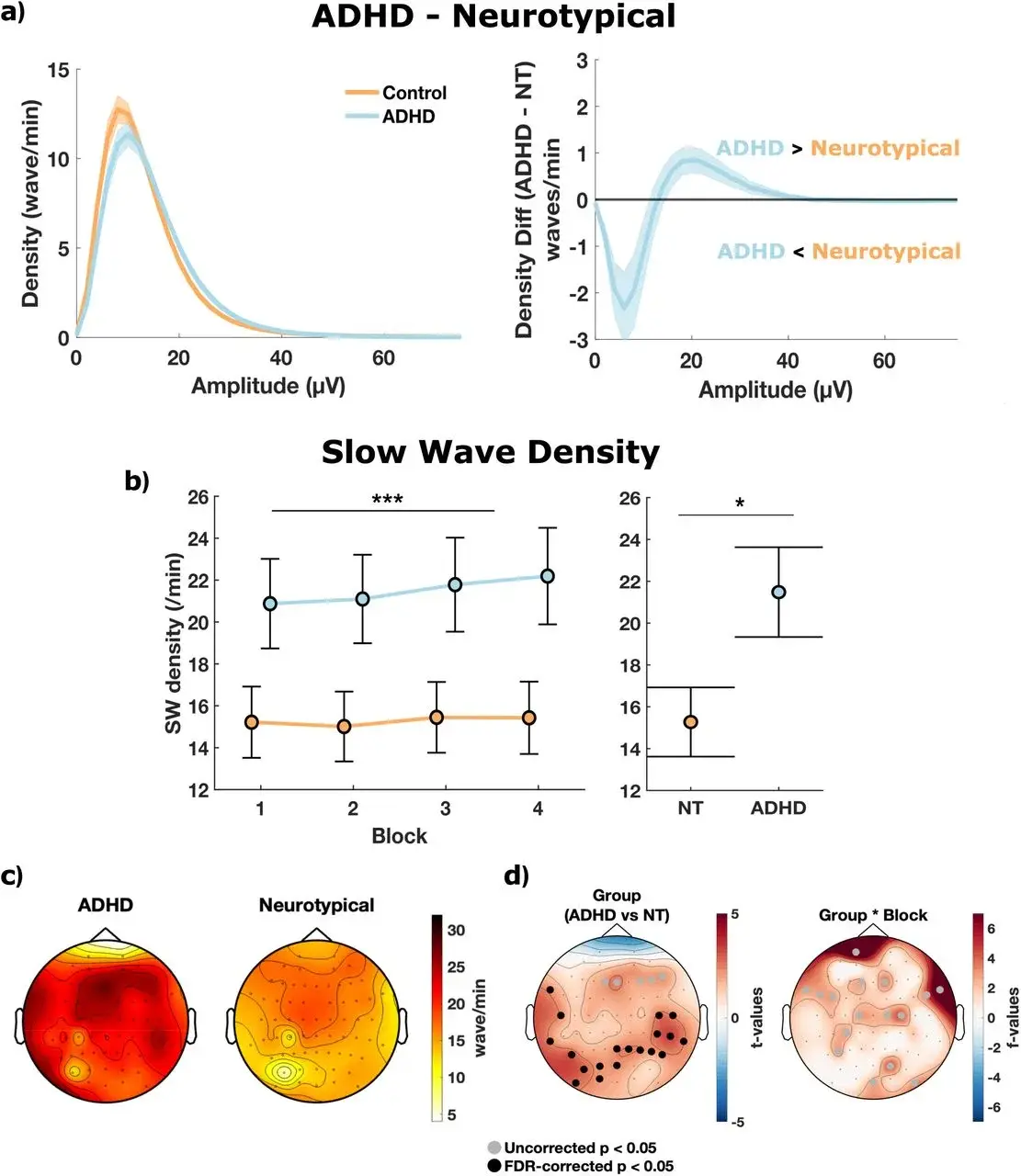
The EEG data told a striking story. Whenever participants reported mind wandering or mind blanking, slow wave density spiked — specifically over parietal and temporal electrodes. These are brain regions involved in sensory integration and spatial awareness. They were dropping into sleep-like states while frontal areas, responsible for motor commands, stayed online.
Local sleep — a phenomenon where isolated cortical areas enter a sleep-like state while the rest of the brain remains awake. First documented in rats in 2011, now confirmed in humans during attention tasks.
This fragmented pattern explains something universally familiar. You drive home on a route you’ve taken a thousand times, arrive safely, and realize you remember nothing from the last twenty minutes. Your motor cortex was steering. Your parietal cortex was napping.
The brain doesn’t switch off like a light. It dims in patches — a rolling brownout rather than a blackout.
ADHD Brains Flicker Far More
Here’s where the data gets sharp. The ADHD group made significantly more commission errors — pressing the button when they shouldn’t have (p = .018). They reported more mind wandering (p = .004) and more mind blanking (p = .016).
But the critical finding was qualitative. In the ADHD group, mind wandering was almost entirely unintentional. The effect size hit d = 0.86 — large by any psychological standard. These participants weren’t choosing to daydream. Their brains were hijacking attention without permission.
ADHD (Attention Deficit Hyperactivity Disorder) — a neurodevelopmental condition affecting 5–7% of children and 2–5% of adults, characterized by difficulties sustaining attention, controlling impulses, and regulating activity levels.
The EEG confirmed this at the neural level. Slow wave density over parieto-temporal electrodes was significantly higher in the ADHD group (cluster-corrected p < .05). More cortical patches were flickering off, more often, and more intensely.
The Chain Reaction: Slow Waves → Attention Collapse
Andrillon’s team went beyond correlations. They ran a mediation analysis — a statistical method that tests whether factor X sits in the causal chain between A and B.
The chain: ADHD → increased slow wave density → more errors and mind wandering. Slow waves weren’t just a side effect. They statistically explained a significant portion of the link between ADHD and attention failures. The brain’s tendency to locally fall asleep was the mechanism driving inattention.
This reframes ADHD. Instead of a vague «attention deficit, ” there’s now a specific neurophysiological marker: the frequency and density of local slow waves during wakefulness. A marker you can measure with an EEG cap and a boring task.
Breaking the Stimulant Paradox
Between 70% and 80% of adults with ADHD report chronic sleep problems. Stimulant medications — methylphenidate, amphetamines — help maintain daytime focus but often wreck nighttime sleep. Poor sleep increases next-day slow wave intrusions. More intrusions mean worse attention. More stimulants. Worse sleep. The cycle tightens.
Andrillon’s finding cracks this loop at a specific point: daytime slow waves themselves. If those waves can be suppressed directly — through neurofeedback, transcranial stimulation, or improved sleep hygiene — attention should improve without escalating the stimulant dose. Not through willpower. Through physics.
For people without ADHD, the takeaway is equally concrete. That afternoon slump where you read the same paragraph four times? Your cortex is literally asleep in spots. The fix isn’t more caffeine. It’s a genuine break — movement, a change of scenery, anything that jolts parietal regions back online.
Where the Evidence Ends
This study was published in the Journal of Neuroscience on March 16, 2026 and has undergone peer review.
Sixty-three participants is standard for EEG research but modest for sweeping claims. The sample skewed about 70% female, limiting generalizability. Critically, the researchers didn’t measure participants’ sleep quality the night before — a single bad night could amplify slow wave intrusions regardless of ADHD status.
The study’s methodological strength lies in combining subjective experience sampling with objective EEG measures. Most attention research relies on one or the other. The mediation analysis adds rigor, though it cannot prove causation — only intervention studies can do that. Whether pharmacological, neurofeedback-based, or sleep-targeted interventions actually reduce these slow waves remains an open question.
There’s also a deeper puzzle. What triggers local sleep in the first place? Is it adenosine buildup? Default mode network dynamics? Simple cortical fatigue from repetitive input? The paper doesn’t answer this — and the answer matters enormously for any future treatment.
What Comes Next
The logical follow-up is a longitudinal design: tracking slow wave density in the same individuals across conditions — after sleep deprivation, after stimulant use, after meditation training. Andrillon’s group has signaled interest in exactly this direction.
If local sleep proves to be a reliable ADHD biomarker, diagnosis could shift from questionnaires and clinical interviews to 20 minutes of a boring task plus an EEG cap. Objective. Reproducible. No subjective judgment calls.
And for everyone else — next time you catch yourself staring blankly at a screen, unable to absorb a single word, know this: your brain isn’t being lazy. Chunks of it are genuinely asleep. The kindest thing you can do is let it wake up.
Frequently Asked Questions
Is it dangerous that my brain «falls asleep» while driving?
Local sleep is one explanation for «highway hypnosis» — driving long distances on autopilot with no memory of the journey. Motor regions keep operating, but cortical areas responsible for conscious awareness can temporarily go offline. Reaction times to unexpected events suffer significantly. Neuroscience-backed driving guidelines recommending breaks every 90–120 minutes aren’t arbitrary — they reflect how long the brain can sustain uniform wakefulness during monotonous input.
Do ADHD medications reduce these brain «flickers»?
This specific study didn’t test medication effects. However, stimulants like methylphenidate and amphetamines increase dopamine and norepinephrine — neurotransmitters that promote cortical wakefulness. In theory, this should reduce local slow wave intrusions. The paradox: stimulants can impair nighttime sleep, and poor sleep increases the very slow waves they’re meant to suppress. Optimizing sleep quality alongside medication may be the key — but direct evidence is still missing.
Can I detect these «flickers» without an EEG?
You’ve probably already noticed them. Re-reading the same sentence three times without absorbing it. Realizing you’ve been staring at a screen for two minutes with zero comprehension. Participants in the study described this as «mind blanking» — a distinct state from active daydreaming. If these episodes are frequent and interfere with daily functioning, it may be worth discussing with a neurologist, especially if other ADHD symptoms are present.
Does this happen to everyone, or only people with ADHD?
Everyone experiences local sleep to some degree during monotonous tasks — the control group showed it too. The difference is magnitude: ADHD brains showed significantly higher slow wave density and more frequent involuntary lapses. Think of it as a volume knob rather than an on/off switch. Most people can push through a boring meeting with effort. For someone with ADHD, that effort may be fighting against cortical regions that are physically asleep.
Could better sleep at night prevent daytime brain «flickers»?
Almost certainly, though this study didn’t directly test it. Sleep research consistently shows that even mild sleep deprivation increases slow wave intrusions during waking hours. For people with ADHD — who already have elevated daytime slow waves — poor sleep likely compounds the problem. Prioritizing sleep hygiene (consistent schedule, dark room, no screens before bed) may be the most accessible intervention, potentially reducing the need for higher stimulant doses.
References
Original
Related Articles
Sleep Loss Turns Gut Against Your Brain
Gut bacteria from sleep-deprived mice triggered Alzheimer's-like tau damage in healthy brains. Scientists traced the full molecular chain.
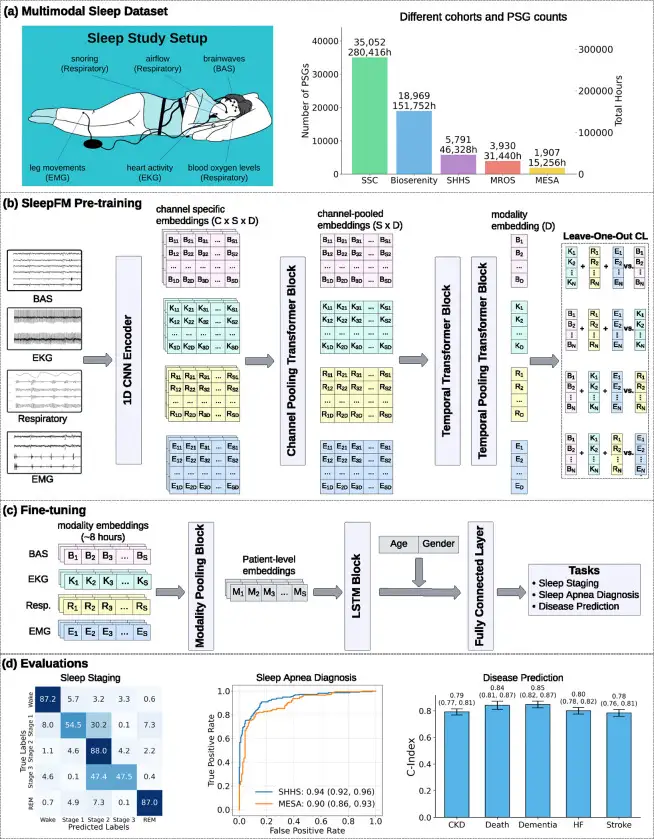
SleepFM: 130 Diseases From One Night of Sleep
Stanford trained a neural network on 585,000 hours of sleep data. It detects Parkinson's, dementia, and cancer years before symptoms appear.
Your Liver, Not Your Legs, May Shield Your Brain From Alzheimer's
UCSF scientists found that a liver enzyme called GPLD1, released during exercise, repairs the blood-brain barrier and reverses memory loss in Alzheimer's mice.