Bacteria That Eat Tumors: Cancer Therapy Breakthrough
Authors: Sara Sadr, Bahram Zargar, Marc G. Aucoin, Brian Ingalls
In 1891, a New York surgeon named William Coley did something his colleagues considered insane: he deliberately injected live bacteria into a patient’s inoperable tumor. Several patients saw their cancers shrink. Nobody understood why.
135 years later, engineers at Canada’s University of Waterloo have finally turned Coley’s wild gamble into a controllable technology. They engineered the soil bacterium Clostridium sporogenes to survive oxygen exposure and — crucially — equipped it with a molecular «group chat»: a quorum sensing system that lets bacteria collectively decide when to strike.
The Craziest Experiment in Oncology History
William Coley had no idea why his approach sometimes worked. He had simply noticed a pattern: cancer patients who developed severe bacterial infections near their tumors occasionally went into remission. Between 1891 and 1936, Coley systematically injected patients with bacterial mixtures — «Coley’s toxins» — documenting dozens of responses.
Coley’s toxins A mixture of heat-killed Streptococcus pyogenes and Serratia marcescens, injected into cancer patients by William Coley from 1891 to 1936. Considered the first form of cancer immunotherapy — decades before the term existed.
His method was crude, unpredictable, and often dangerous. When radiation therapy and chemotherapy emerged in the mid-20th century, Coley’s approach was abandoned. But the idea that bacteria could destroy tumors never truly died — it went underground.
Uninvited Guests Inside Tumors
In the 1950s, researchers discovered something remarkable: Clostridium spores, when injected intravenously, traveled through the body but germinated in only one place — inside solid tumors.
The reason lies in tumor physics. When a malignant tumor outgrows its blood supply, its center becomes a dead zone — the necrotic core. No oxygen, no immune cells, almost no nutrients. For most organisms, this is a wasteland. For Clostridium, it’s paradise.
Necrotic core An oxygen-free zone at the center of large solid tumors where cells have died due to insufficient blood supply. Unreachable by most drugs and immune cells — a key reason why many tumors recur after treatment.
Clostridium species are strict anaerobes. They don’t just survive without oxygen — they require its absence. Oxygen is lethal to them. A tumor’s necrotic core is their ideal habitat.
This created an astonishingly precise drug delivery address: bacteria would autonomously navigate to the very region that chemotherapy and immune cells cannot reach. The perfect weapon — in theory. In practice, serious problems awaited.
Clinical Trials: Promise Meets Sepsis
In the 1990s, researchers developed CDEPT — Clostridium-directed enzyme prodrug therapy. The concept was elegant: bacteria deliver an enzyme to the tumor, then a harmless prodrug is administered. The enzyme converts the prodrug into a cytotoxin — but only at the tumor site. Targeted chemotherapy activated by a living courier.
CDEPT (Clostridium-directed enzyme prodrug therapy) A strategy where engineered Clostridium bacteria deliver a specific enzyme to tumors. The patient then receives an inactive prodrug that only this enzyme can convert into a toxin — confining cell death to the tumor.
In the 2010s, Shibin Zhou’s team at Johns Hopkins brought bacterial cancer therapy into clinical trials using C. novyi-NT — a strain stripped of its primary lethal toxin. In Phase I/Ib trials (NCT01924689), spores were injected directly into tumors of 24 patients.
The results were simultaneously encouraging and sobering. Spores germinated successfully in 42–46% of patients. Disease stabilization was observed in 86%. But several patients developed severe complications: gas gangrene, sepsis, abscesses.
The fundamental problem was that the bacteria remained «dumb.» They couldn’t sense when their numbers were sufficient for an effective attack. They couldn’t stop growing after the tumor was destroyed. They couldn’t adapt to mixed oxygen environments within the tumor. Powerful biological weapons — but without a safety switch.
Two Keys from Waterloo
The University of Waterloo team, led by Bahram Zargar and Brian Ingalls, took a different approach. Instead of C. novyi-NT, they chose C. sporogenes — a related species easier to engineer in the lab. Their goal: make the bacterium not just effective, but intelligent.
The first key came in 2023. Graduate student Sara Sadr and colleagues inserted an oxygen-tolerance gene borrowed from a related microorganism, allowing C. sporogenes to temporarily survive oxygen exposure. This solved a critical logistics problem: when injected intravenously, spores must pass through oxygen-rich blood and tissues before reaching the tumor. Without protection, they would die en route.
The second key — published in December 2025 in ACS Synthetic Biology — was even more elegant.
Bacterial Democracy
Imagine you’re a bacterium. You’ve found a tumor’s necrotic core and started multiplying. But how do you know when there are enough of you to mount an effective attack? When is it time to switch on «combat mode»?
This is where quorum sensing enters the picture — one of nature’s most sophisticated communication systems.
Quorum sensing A system of «bacterial democracy»: cells continuously release signaling molecules. When the concentration crosses a threshold (meaning enough «voters» are nearby), collective behavior changes are triggered — such as activating virulence genes, producing bioluminescence, or forming biofilms.
Sadr and colleagues transplanted the agr quorum sensing system from Staphylococcus aureus — well-characterized and reliable — into C. sporogenes. Think of it as giving bacteria a group chat with a voting threshold.
Each bacterium continuously releases a small signaling peptide called AIP (autoinducing peptide). When few bacteria are present, AIP concentration stays low — nothing happens. The bacteria live, multiply, but stay silent. When the population reaches critical mass, the signal crosses the threshold — and target genes switch on simultaneously across the entire colony.
In this study, the researchers used GFP (green fluorescent protein) as a reporter gene rather than a therapeutic payload. It’s a proof-of-concept indicator: if bacteria glow green, quorum sensing works. And it did. At threshold population density, C. sporogenes collectively activated GFP — confirming that the heterologous agr system functions in its new host.
A Living Programmable Weapon
Why does this matter beyond basic science? Because quorum sensing is a built-in safety mechanism.
Remember the core problem in C. novyi-NT trials: uncontrolled bacterial growth leading to sepsis. Now imagine a system where the therapeutic gene — whether an enzyme for CDEPT, a tumor-destroying cytokine, or a direct toxin — activates only at sufficient bacterial density. Meaning only deep inside the tumor.
If a single bacterium lands in healthy tissue? Nothing happens. No quorum, no activation, no danger. This is a fundamentally different safety profile.
Combined with oxygen tolerance, the complete platform works like this:
- Spores are injected intravenously — the oxygen-tolerance gene protects them during transit
- Spores find the tumor — they germinate exclusively in the anaerobic necrotic core
- Bacteria multiply — continuously «voting» via quorum sensing
- Quorum is reached — the therapeutic gene activates
- The tumor is destroyed from within
The therapeutic «warhead» is interchangeable: enzymes for prodrug activation, cytokines for immune stimulation, direct toxins for cell killing. A modular platform — like biological Lego.
Critical Assessment
The Waterloo study is a proof of concept, not a finished drug — and the gap between the two is wide.
What makes this work genuinely significant is its position in the field’s history. Successfully transplanting the agr quorum sensing system into C. sporogenes is a species-level first — nobody has done it before — and the choice of agr is not arbitrary: decades of S. aureus research have produced an unusually complete characterization of how this circuit behaves, which reduces the risk of unpredictable emergent effects. The modular design philosophy deserves equal credit: using GFP as a reporter gene rather than a therapeutic payload is a deliberate act of scientific restraint, making the system easy to verify and easy to extend. That the team has now published two methodically connected papers, in 2023 and 2025, building one capability on top of another, signals a disciplined long-game strategy rather than a one-shot publication play.
The limitations, however, are not minor caveats — they define how far the technology actually is from a patient. The oxygen-tolerance modification and the quorum sensing circuit exist in separate strains and have not yet been combined into a single organism. Every experiment to date has been conducted in vitro, meaning no animal data exists at all; the immune system’s reaction to engineered Clostridium colonizing a tumor is entirely unknown. And evolutionary stability is a genuine concern: bacterial populations replicate fast and can shed foreign genetic material over generations, so whether the inserted circuits will remain intact under selection pressure inside a living host is an open question the current work cannot answer.
Deeper uncertainties lie beyond the laboratory. What happens to the bacteria once the tumor is destroyed and oxygen floods the former necrotic zone — will they die naturally, or will a synthetic «kill switch» be required? What is the minimum tumor size needed to form a necrotic core large enough for reliable colonization? And perhaps most unsettling from a biosafety perspective: could horizontal gene transfer propagate the engineered capabilities to other microorganisms in the gut or the environment? None of these questions are hypothetical obstacles; they are the actual milestones that stand between this result and a clinical trial.
This analysis is based on a peer-reviewed publication in ACS Synthetic Biology (IF ~5.0). The paper describes early-stage in vitro work. Years of additional research are needed before clinical application.
What Comes Next
The Waterloo team’s immediate next step is combining both modifications in a single C. sporogenes strain and replacing the GFP reporter with a therapeutic gene. Then: pre-clinical testing in mouse models of solid tumors.
Meanwhile, several other bacterial cancer therapy programs are advancing worldwide. C. novyi-NT from BioMed Valley Discoveries remains the most advanced candidate (Phase II, NCT03435952). Synlogic is engineering E. coli for immuno-oncology applications. In 2023, researchers at Fred Hutchinson described novel methods for investigating tumor necrotic cores.
Realistic timeline: 5–10 years before clinical trials begin for this specific platform. But if pre-clinical results are strong, this could become one of the most significant oncology breakthroughs of the 21st century.
135 years ago, William Coley injected bacteria into tumors blindly, hoping for a miracle. Now engineers are building bacteria that decide for themselves when and where to strike. From one surgeon’s desperate experiment to programmable living medicine. The road has been long — but we may finally be approaching the destination.
Frequently Asked Questions
What happens to the bacteria after the tumor is destroyed?
C. sporogenes is a strict anaerobe — it dies in the presence of oxygen. When the necrotic core collapses and oxygenated blood flows into the area, the bacteria should die naturally. However, researchers are also developing genetic «kill switches» — engineered self-destruct mechanisms triggered by an external signal — as an additional safety layer.
Could the bacteria mutate and become dangerous?
Theoretically, yes — like any living organism. But the quorum sensing system provides a safety layer: even if a mutation alters one bacterium, it needs a critical mass of cooperating bacteria to activate dangerous genes. Additionally, engineers plan to use auxotrophic strains — bacteria unable to synthesize essential amino acids, meaning they cannot survive without artificial supplementation.
How is this different from cancer immunotherapy?
Immunotherapy (such as checkpoint inhibitors) «unblocks» the patient’s own immune system to fight cancer. But it works poorly against solid tumors with necrotic cores — immune cells simply cannot reach those oxygen-free zones. Bacterial therapy operates precisely where other approaches fail. Ideally, both methods would complement each other for a more complete response.
When could this become available to patients?
The Waterloo team is at the in vitro stage — clinical trials are likely 5–10 years away. However, other bacterial therapies are already in clinical testing: C. novyi-NT is in Phase II trials (NCT03435952). If the Waterloo platform shows strong pre-clinical results, the timeline could accelerate significantly.
Which types of cancer could benefit from this approach?
Theoretically, any solid tumor with a pronounced necrotic core. The most likely candidates include glioblastoma (brain cancer), pancreatic cancer, and sarcomas — tumors that respond poorly to standard therapy and frequently develop extensive oxygen-free zones. Liquid cancers (leukemias, lymphomas) are not suitable for this approach, as they don’t form solid masses with necrotic cores.
References
Original
Related
Related Articles
A Bacterium Frozen for 5,000 Years Is Resistant to 10 Antibiotics — and May Help Discover New Ones
A 5,000-year-old microbe from a Romanian ice cave resists 10 modern antibiotics. Its genes may hold keys to discovering new drugs.
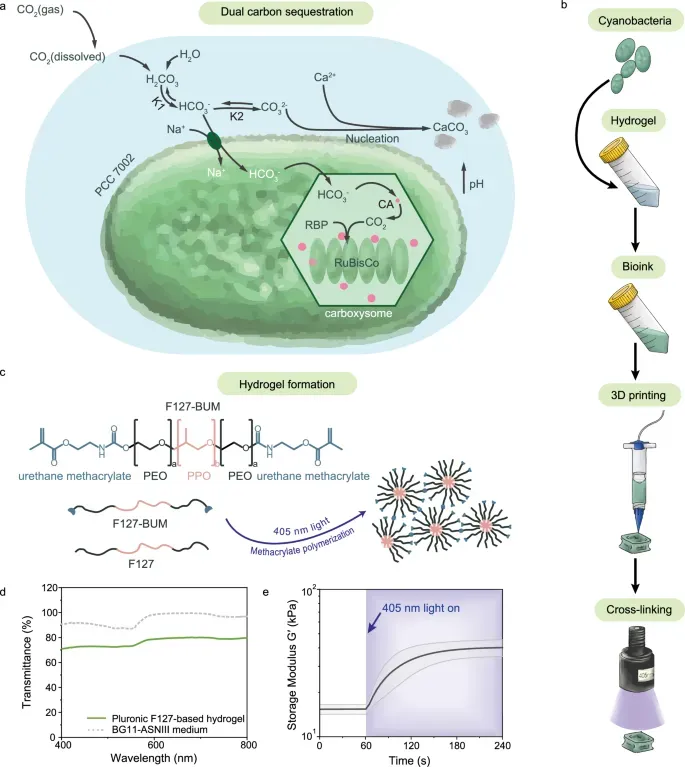
Living Buildings: 3D-Printed Hydrogel with Bacteria Captures CO₂
ETH Zurich engineers created a 3D-printable hydrogel with cyanobacteria that captures carbon dioxide through dual mechanisms — photosynthesis and carbonate mineralization.
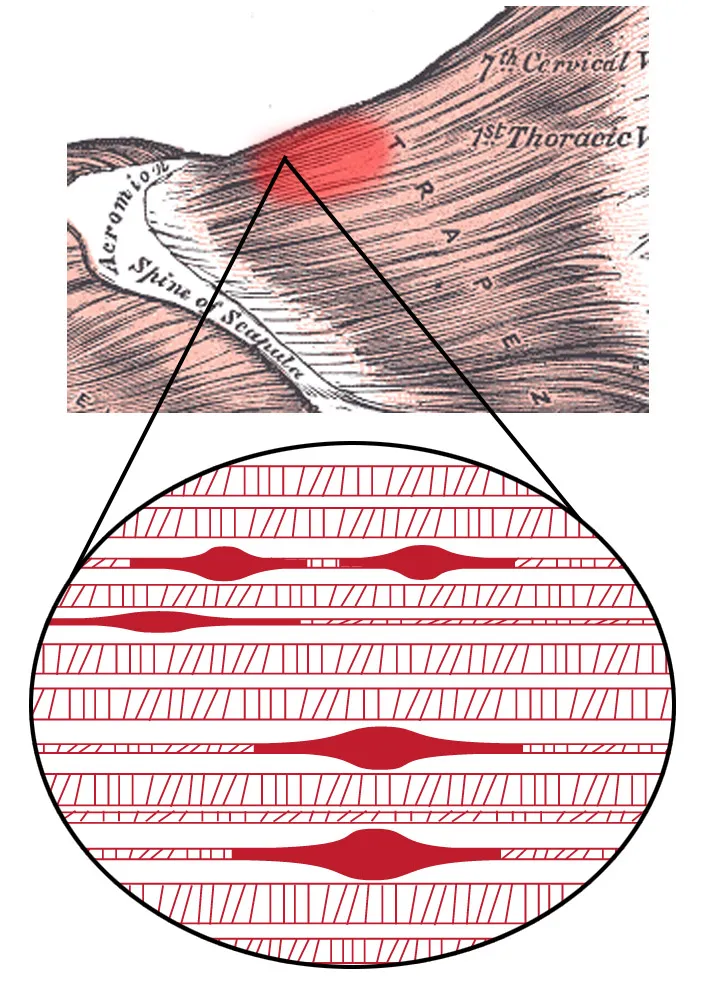
Muscle Knots Explained: The 2024 Trigger Point Breakthrough
Why muscle knots form, how they harm your health, and how to treat them — featuring the molecular pathway discovered in 2024.